DR MIKE YEADON COVID DEATH SHOTS IS MEDICAL MURDER
Dr Mike Yeadon – Let’s not put a Gloss on It. The COVID death shots Is Medical Murder
First RSV Emergency Declared as Pfizer and GSK Race to Get Vaccines Approved

By Michael Nevradakis, Ph.D. | The Defender | November 8, 2022
Southern California’s Orange County Health Department this month declared a local health emergency over concerns around the rising number of pediatric cases of Respiratory Syncytial Virus (RSV).
County officials also declared a Proclamation of Local Emergency, allowing them to access state and federal resources in response to the spread of RSV.
The news came amid media warnings of a looming “tripledemic” of RSV, influenza and COVID-19 and news that Pfizer and GlaxoSmithKline (GSK) are close to securing regulatory approval for their RSV vaccine candidates — including Pfizer’s RSV vaccine for pregnant women.
RSV was first identified in 1956, and frequently affects children, with a majority of childhood cases occurring before age 2. For most children, symptoms are similar to those of the common cold, according to the Centers for Disease Control and Prevention (CDC).
It can, however, cause serious symptoms in the very young and the elderly, although childhood deaths are infrequent, according to CDC data.
According to California attorney Rita Barnett-Rose, there’s no evidence to support claims that Orange County hospitals are overextended, or to justify a state of emergency.
In an interview with The Defender, Barnett-Rose drew parallels between Orange County’s emergency declaration for RSV and other state, local and federal emergency declarations for COVID-19:
“One of the other alarming things that happened with COVID is when [Gov. Gavin] Newsom declared the state of emergency in COVID. He also waived the 30 and 60-day review periods for all local governing boards.
“So, yeah, so that’s what everyone is concerned about, this perpetual state of emergency.
“And the other thing that it triggers is it asks, once they declare a state of emergency, aside from getting the funding, it also triggers a request to Governor Newsom to declare a statewide state of emergency and then also to have Newsom ask President Biden to declare a national state of emergency.”
That could lead to Emergency Use Authorization (EUA) of RSV vaccines, according to Barnett-Rose, who told The Defender there may be an avenue to challenge the legality of the latest Orange County emergency declaration.
Is pharma using the COVID playbook to push RSV vaccines?
Dr. Regina Chinsio-Kwong, Orange County’s health officer, said in a press release county officials issued the declaration due to “rapidly spreading virus infections causing record numbers of pediatric hospitalizations and daily emergency room visits.”
The declaration will allow the county to access state and federal resources and to allow non-pediatric hospitals to offer beds to sick children, Chinsio-Kwong said.
Children’s Health Defense (CHD) California Chapter notes that this declaration was unanimously ratified “over the objections by members of the public who showed up to speak against the declarations.”
Dr. Melaine Patterson, chief nursing officer for Children’s Health Orange County, said area hospitals are seeing an “unprecedented” volume of children visiting their emergency rooms — approximately 400 per day, with wait times of “four to 12 hours.”
She also said that, as of Nov. 1, there were 285 patients in her hospital, compared to 188 on that date last year, adding that 11 of those current patients were placed in ICU as a result of RSV infections. RSV cases also are on the rise nationally.
Chinsio-Kwong advised the public to remain “up to date with other vaccinations such as flu and COVID-19” to “reduce the burden on hospitals this fall and winter.”
“Our best shot at protecting ourselves and our children from respiratory illnesses continues to be the same things we practiced throughout the pandemic including the use of masks when indoors around others and staying home when you are sick,” Chinsio-Kwong said.
However, Barnett-Rose said hospitals in Orange County may be overextended not due to an unusually high number of admissions, but due to unvaccinated staff having been let go.
She told The Defender :
“I do think that has a big role in it, and a lot of people pointed that out during the hearings and public comments were made about that.
“They were trying to claim … there were not enough pediatric experts or pediatric-trained doctors and medical professionals, so they needed to go outside of the county. And I do think a big part of that is that they did let go of many pediatric nurses and pediatric physicians that refused to take the vaccines. So I do think that this is a crisis of their own making … if there is even a crisis.”
San Diego County also issued a warning to its residents Nov. 4 over rising RSV cases but stopped short of declaring an emergency.
CHD California questioned why Orange County would declare a “Local Health Emergency” now — even though local COVID-19 states of emergency haven’t been lifted.
Despite claims that the purpose of these new declarations is exclusively meant to help county hospitals, CHD California said, “the language of the emergency Resolutions says something else entirely”:
“Both the Resolution to ratify the Local Health Emergency under Health & Safety Code section 101080 and the Proclamation of a Local Emergency under Government Code section 8630 declare the need for these local emergencies not only based on the asserted need to handle allegedly rising cases of RSV — but also because of other unnamed ‘other respiratory illnesses’ that were not discussed during the special meeting or otherwise disclosed to the public.”
Barnett-Rose provided insights into the legal meaning underlying these declarations. She told The Defender :
“There’s a local health emergency, which is declared by the public health officer as opposed to the [county] board … and that is when the health officer says … ‘I’ve been talking to the hospitals and we’ve done some assessments and there’s an increasing rate of respiratory illnesses and the children’s hospitals, and so, because of this, I am asking to declare a local health emergency.’ …
“But her authority to declare a local health emergency only lasts for seven days unless it’s ratified by our board of supervisors. And so, on Nov. 2, our board ratified that, which means now it’s in place and it needs to be renewed or looked at every 30 days, unless Governor Newsom waives that condition.”
CHD California argued that the “overly broad language used in these emergency declarations should be deeply concerning to anyone wary of a repeat of the “public health” measures ordered under COVID-19,” noting that adding the vague statement referring to “other respiratory viruses” creates “plenty of room for further abuse.”
Barnett-Rose told The Defender that, during the Nov. 2 meeting, Chinsio-Kwong said, “No restrictions are currently [emphasis added] in place,” a statement that “caused … a little bit of an uproar during the hearing because of her wording, which suggests that indeed they may end up rolling out mask mandates or some other mandates.”
Under this type of “slippery language,” argued CHD California, even the common cold could be used to issue an emergency declaration.
Barnett-Rose agreed:
“How do you measure that? How do you dispute it? How do you examine it? How do you end it?
“If you’re going by one infectious disease, at least you can try to find some data on hospitalizations or cases or deaths or ICU beds. But when it’s everything, it can still include COVID, flu, cold, COPD, a whole bunch of things that would qualify as respiratory illnesses.”
Barnett-Rose said that attempts will be made to legally challenge these declarations as “overbroad,” noting, however, that while case law precedent exists to challenge overbroad legislation, there is no such precedent in place for states of emergency.
Barnett-Rose she’s looking into whether there’s some way to litigate against these broad states of emergency.
When statutes are overbroad, she said, sometimes you can challenge them for being overley broad or too vague, but she’s not sure if there’s a similar precedent for challenging an overly broad state of emergency.
Declaring a local state of emergency at the county level could trigger a chain of events leading all the way to a national declaration of emergency, according to CHD California:
“By ratifying and/or declaring these local emergencies, the Board has now triggered an official request to Governor Newsom that he also declare a state-wide state of emergency on the same overly broad basis — and to ask the President of the United States to declare a national state of emergency.”
“A nationally declared state of emergency may trigger all of the same measures that have already deeply divided our country: masks on airplanes, business and school closures, and coercive school and employee mandates for experimental ‘vaccines’ offered as the ‘only solution’ back to Freedom,” CHD California argued.
Barnett-Rose said the emergency declaration in Orange County may be tied to the fact that there are multiple RSV vaccines already in the pipeline:
“The reason this is becoming really huge news is because this is what happened with the COVID state of emergency, too, it kind of went like that in dominoes.
“Once Biden declares a national state of emergency, then that triggers all of the emergency use authorizations that allowed the so-called vaccines for COVID. And I’m sure you already know that there’s vaccines in the works now for RSV that they’re floating out there in the news as well.”
Recounting events that occurred in relation to COVID-19, Barnett-Rose told The Defender that Orange County and some other counties in California declared a state of emergency, “then it went to the state, then it went national, and then, of course, once there’s a national emergency, you can go into Iowa.”
“So I do think that this is very suspicious timing, the articles that we are seeing in the news about being close to this RSV vaccine,” Barnett-Rose said. “And so, I do think that they’re trying to repeat the same playbook.”
Up to 30 RSV vaccine candidates already in pipeline
Pharmaceutical industry publication FiercePharma reported that Pfizer and GSK “moved closer to regulatory approvals” in the past week, and “could each see the first-ever approvals for an RSV vaccine in 2023.”
GSK’s candidate will be reviewed May 3, 2023, while Pfizer is “eyeing an approval filing by year-end.”
In all, up to 30 RSV candidate vaccines have been identified by the National Institutes of Health (NIH).
GSK has secured priority review status for its RSV vaccine following the submission of data from its AReSVi-006 phase 3 trial. The candidate vaccine exclusively targets adults 60 and older, and the data reportedly “showed high overall vaccine efficacy against RSV lower respiratory disease” at a rate of 82.6%, Fierce Biotech reported.
GSK initially intended to develop an RSV vaccine for older adults and for infants, but plans for the latter were shelved due to a “safety concern.” Nevertheless, GSK CEO Emma Walmsley in a call last week with investors described GSK’s new vaccine as “a very significant scientific achievement.”
Previous efforts to develop an RSV vaccine have failed because they “had a persistent tendency to cause worse disease,” due to “a phenomenon known as antibody-dependent enhancement (ADE).”
Dr. Ruth Karron, a pediatrician and professor of international health at the Johns Hopkins Bloomberg School of Public Health, said, “The difference this year is the general public for the first time is probably eagerly awaiting an RSV vaccine, because they’re seeing firsthand just how much of a problem RSV can be,” in a statement drawing a clear connection with the reported increase in RSV cases this year.
Pfizer announced Nov. 1 that its own candidate RSV vaccine wrapped up its phase 3 trial for efficacy early. Unlike GSK’s candidate vaccine, Pfizer’s vaccine targets pregnant women. The vaccine is designed to be administered early in the third trimester with the intent that the vaccine-induced antibodies be passed from the mother to her newborn.
Pfizer, in its Nov. 1 announcement, claimed that its bivalent RSV prefusion vaccine candidate RSVpreF, also known as PF-06928316, delivered “broadly positive” results — namely, a significant reduction in the rate of respiratory illness in newborns and their mothers.
Pfizer claimed 81.8% efficacy against severe cases of RSV for babies up to three months, and 69.4% efficacy after six months.
Pfizer’s MATISSE (MATernal Immunization Study for Safety and Efficacy) trial involved 7,400 pregnant women and their newborns, tracking the infants for one year following birth, and some for two years. It was conducted in 18 countries beginning in June 2020.
Pfizer’s vaccine trial did not deliver statistically significant results regarding the prevention of infant medical visits caused by RSV, including for non-severe cases.
Nevertheless, Pfizer said there are “no major safety concerns” with its product, adding:
“At the recommendation of the DMC [Data Monitoring Committee], and in consultation with the U.S. Food and Drug Administration (FDA), Pfizer has stopped enrollment in the study.
“Based on these positive results Pfizer plans to submit a Biologics License Application (BLA) to the FDA by the end of 2022 for the vaccine candidate followed by other regulatory authorities in the coming months.”
Pfizer’s phase 3 trial data has not yet been vetted by independent experts, and has not been peer-reviewed or published.
On March 2, 2022, Pfizer’s candidate vaccine received a Breakthrough Therapy Designation from the FDA, while previously, in November 2018, the FDA granted it Fast-Track status, according to the Pfizer release.
“Pfizer’s investigational RSV vaccine candidate builds on foundational basic science discoveries including those made at the National Institutes of Health (NIH),” the vaccine maker said in its press release.
The NIH research in question also was “key to the hugely successful COVID-19 vaccines,” according to ABC 7 Los Angeles.
Others looking to develop an RSV vaccine include Moderna and AstraZeneca and Sanofi, in a joint initiative. Moderna received fast-track designation from the FDA on Aug. 3, 2021, for its mRNA-1345 RSV vaccine for older adults, while AstraZeneca and Sanofi are developing a viral vector RSV vaccine by the name of “nirsevimab.”
An August 2021 article in The Defender predicted that RSV would represent the “new frontier for vaccine development,” as pharmaceutical companies would be “lured by the prospect of a large untapped global RSV vaccine market,” leading to a vaccine development “gold rush.”
Media tout benefits, ignore safety concerns
Online news portal Vox described the development of new RSV vaccines as “very, very good news” in a recent article.
Coupled with media coverage about “rising” RSV cases nationwide, it appears the stage is being set to create an emergency in the public consciousness, which could lead to EUAs being granted to the new vaccines and a rush to get the public vaccinated.
Blogger James Roguski, however, called into question the results of the Pfizer phase 3 trial, raising many safety concerns that are indicated in the company’s own data but were downplayed by Pfizer in its public proclamations. In a recent post, he wrote:
“In the Phase II study, researchers found that women who received RSVpreF vaccine containing aluminum hydroxide had a higher incidence of local reactions than those who received RSVpreF vaccine without aluminum hydroxide.
“It seems like 1 of the 117 infants in the placebo group suffered fetal death and 6 infants in the ‘vaccinated’ group are unaccounted for.”
Roguski also highlighted the many adverse events that were recorded for both infants and pregnant mothers who participated in the study, none of which were recorded for the placebo group.
For infants, some of these adverse events included acute respiratory failure, bronchiolitis, cardiac murmur, chordee (bent penis), conjunctival hemorrhage, hypoglycemia, jaundice, low birth weight, neonatal hypoxia, neonatal respiratory failure, seizure, sepsis, upper respiratory tract infection and vascular malformation.
Pregnant women, in turn, experienced adverse events including fetal growth restriction, fetal tachycardia, gestational diabetes, nonreassuring fetal heart rate pattern, premature labor, premature separation of placenta, vaginal hemorrhage and thrombocytopenia.
For some, these safety concerns are conjuring up memories of previously failed attempts to develop an RSV vaccine.
A vaccine tested in 1966 led to the deaths of two toddlers as a result of “enhanced disease symptoms,” while many infants “suffered worse symptoms than usual, and needed to be hospitalized.”
As previously reported by The Defender, RSV itself originated in monkeys housed in a Maryland facility where they were used to conduct polio vaccine research. Remarking on this and the connection to RSV vaccine development today, Brian Hooker, CHD’s chief scientific officer, told The Defender, “It is incredible that the vaccine industry can create a disease — RSV from the polio vaccine — and then create another vaccine to ‘prevent’ that disease. Talk about a business proposition!”
Michael Nevradakis, Ph.D., based in Athens, Greece, is a senior reporter for The Defender and part of the rotation of hosts for CHD.TV’s “Good Morning CHD.”
This article was originally published by The Defender — Children’s Health Defense’s News & Views Website under Creative Commons license CC BY-NC-ND 4.0. Please consider subscribing to The Defender or donating to Children’s Health Defense.
Dr. Clare Craig On The Three Key Factors Of The Pandemic
The Fat Emperor – Ivor Cummins | November 9, 2022
Dr. Clare Craig is one of the best pathologists out there, and deeply understands the scientific details of the pandemic – and I caught her in person at the International health congress in Portugal – all scientific talks are here or will be uploaded shortly https://odysee.com/@MartaGB:2
Department of Homeland Security CISA
US Government Office of Medical Censorship and Propaganda
By Peter A. McCullough, MD, MPH | Courageous Discourse | November 11, 2022
The US Department of Homeland Security (HSA) is conducting medical censorship while hiding in plain sight. The website for the Cybersecurity and Infrastructure Security Agency (CISA) has resources to engage vigilante “disinformation” police to assist HSA in their mission of silencing opinions on COVID-19 and pandemic response. The main stated target is disinformation defined as information deliberately created to mislead, harm, or manipulate a person, social group, organization, or country. Their toolkit allows any user to use “products” and tailor them with official logos to spread the government propagandized message:[i]
“COVID-19 DISINFORMATION TOOLKIT
These Toolkit resources are designed to help State, local, tribal and territorial (SLTT) officials bring awareness to misinformation, disinformation, and conspiracy theories appearing online related to COVID-19’s origin, scale, government response, prevention and treatment. Each product was designed to be tailored with local government websites and logos.
Download and share these resources—talking points, FAQs, outreach graphics, and posters—to help spread awareness.”

The toolkit directs well-intended users to use images, talking points, and documents to deliver a message. There is only ONE source of trusted information — you guessed it — state and local agencies who rely upon the CDC!

So, the picture is becoming more clear on how the US government operationalized a propaganda campaign on its own people from the very beginning of the COVID-19 crisis. They took these steps:
1) establish a single source of truth — the CDC,
2) weaponize CISA to declare “disinformation” their target,
3) enlist a legion of volunteer deputies without any official authority or accountability to operate within social media and all walks of life, giving public service messages telling Americans the CDC is the only trusted source of information. The converse of this assertion–anything else must be considered untrue and up for being nailed as “misinformation,” “disinformation,” or “malinformation.”
Don’t be surprised if FOIA-obtained documents demonstrate CISA and CDC were operating as partners in established campaigns with social media, mainstream television, print media, corporations, schools, and every aspect of life. Nothing can be more dangerous to public health. Directing all trust to a single source of medical information that is not contemporary, has no regular schedule of review or public briefings, is not transparent with data (e.g., the withheld V-Safe dataset), and has woefully lagged on major scientific developments (contagion control, testing, vaccine safety).
It’s a mind-blowing reality that our government agencies, in a planned and coordinated manner, have operationalized a plan to control information and spread propaganda in order to influence behavior. They pitted agencies against citizens and individuals against one another and set social media as the main battleground. The CDC and DHS CISA should be prime targets of US Senate and Congressional Investigations into our disastrous pandemic response.
[i] DHS CISA Publication: “We’re in This Together. Disinformation Stops With You.”
Good News on Omicron Outcomes from Prison
Captive Environment Allows Real Look at Risk for Hospitalization and Death
By Peter A. McCullough, MD, MPH | Courageous Discourse | November 8, 2022
Prisoners and staff are a prototypical congregate group amenable to epidemiologic study and in the setting of the SARS-CoV-2 outbreak, ideal for study of the patterns of spread and risk for serious outcomes.
Throughout the pandemic, review and interpretation of peer-reviewed manuscripts in widely read journals has required careful review of data in supplementary tables. The conclusions by the authors in many papers must be ignored given the heavy bias for all journals to “promote” mass vaccination. For example, thus far, there has not been a single paper in the New England Journal of Medicine that has concluded COVID-19 vaccines are unsafe, or not indicated, or do not have a favorable risk to benefit ratio.
Prior to COVID-19 for any new therapy or surgery, there are always divergent views where some papers conclude an intervention is beneficial and others do not. Disagreement is normal in medicine. The pervasive heavy-handed government intelligence community and public health agency operations have biased the medical journals to publish a one-sided story on COVID-19 vaccination. The paper by Chin et al, in the New England Journal of Medicine is a perfect example.[i] They studied 59,794 residents and 16,572 staff, using data collected from December 24, 2021, through April 14, 2022. Buried in the supplemental tables are important data on the real risk of hospitalization and death with the Omicron variant in prisoners characterized by whether they had prior COVID-19 and how many doses of a vaccine they had received.
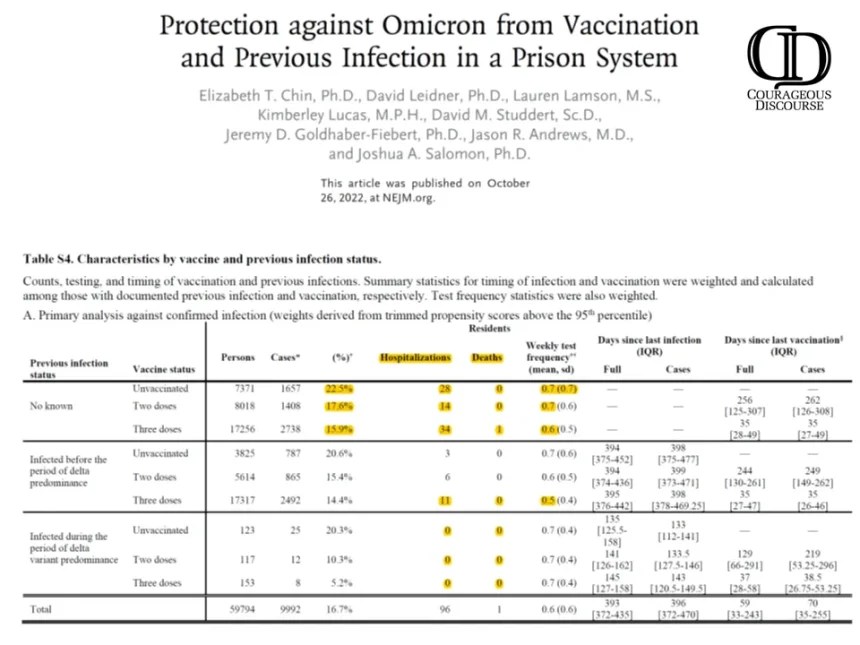
Chin ET, Leidner D, Lamson L, Lucas K, Studdert DM, Goldhaber-Fiebert JD, Andrews JR, Salomon JA. Protection against Omicron from Vaccination and Previous Infection in a Prison System. N Engl J Med. 2022 Oct 26. doi: 10.1056/NEJMoa2207082. Epub ahead of print. PMID: 36286260. Supplemental Tables.
As shown, there is a negligible risk of hospitalization and death with Omicron infection. Specifically, there were zero deaths in the unvaccinated. To demonstrate how far off-base authors and editors are on interpretation, take a look at the paper’s conclusions:
“Our findings in two high-risk populations suggest that mRNA vaccination and previous infection were effective against omicron infection, with lower estimates among those infected before the period of delta predominance. Three vaccine doses offered significantly more protection than two doses, including among previously infected persons.”
My conclusions are considering natural immunity status, COVID-19 vaccination provided no meaningful protection against hospitalization and death with Omicron. Based on these data, a rational prison physician or health administrator would conclude that bivalent boosters for Omicron are not clinically indicated nor medically necessary. So next time you see headlines regarding a journal article, look for a second opinion from “Courageous Discourse” or other reliable stacks and sources of analysis.
[i] Chin ET, Leidner D, Lamson L, Lucas K, Studdert DM, Goldhaber-Fiebert JD, Andrews JR, Salomon JA. Protection against Omicron from Vaccination and Previous Infection in a Prison System. N Engl J Med. 2022 Oct 26. doi: 10.1056/NEJMoa2207082. Epub ahead of print. PMID: 36286260.
Why Are People Losing Confidence in Vaccines?
BY DR DAVID PATON | THE DAILY SCEPTIC | NOVEMBER 8, 2022
A new peer reviewed article in the journal Vaccine has been published comparing surveys data on attitudes to vaccination from before the pandemic with attitudes now.
The authors report that “paradoxically, despite the success of COVID-19 vaccination campaigns, vaccine confidence has significantly declined since the onset of the pandemic”.
I am not quite sure why the authors appear so surprised at their result but a clue can be found in their use of the word “despite”. In many countries the “success” of the COVID-19 vaccination campaign relied in large part in pressuring, bullying and sometimes coercing people to get vaccinated.
Now if governments tell you that getting vaccinated is in your best interests, but that nonetheless those who choose not to get vaccinated will be pilloried in the press and on social media, barred from participating in normal everyday activities and, in some cases, sacked from their employment, perhaps we should not be surprised that people start to doubt whether those governments really do have their best interests at heart.
And those doubts have substance behind them. From a very early stage, it was clear that for many people, the known risks from vaccination probably outweighed any likely benefit. This was most obvious for those who had already had Covid (and for whom the marginal impact of vaccination in preventing a further infection was small), for groups who faced very low risks of serious illness if they contracted Covid and especially for young males for whom vaccination seems to be bring additional risks of heart problems.
But instead of acknowledging that Covid-19 vaccines may make sense for some people and not everyone, too many public health officials, scientists and politicians have systematically downplayed immunity from previous infection, brushed aside concerns over side effects and dismissed concerns that the roll out to the whole population was rushed given uncertainty over long term effects.
The latter was a particularly serious error. Right from the start of the roll out the public was assured that the vaccines had been thoroughly tested. Yet in May 2021, the Government decided to stop providing the AstraZeneca vaccine to under 40s due to the relatively high rate of blood clots. That decision would have been little consolation to the families of the 73 people whose deaths the MHRA report as being linked to that vaccine.
Rather than taking that as a lesson, authorities around the world doubled down and rolled out vaccine mandates, passports and travel restrictions as a way of twisting more arms into receiving the jab. Ironically, these measures were aimed primarily at young people for whom the benefits of vaccination were lowest and (particularly for males) the risks seem highest.
The official rationale for these policies was that vaccination would help protect others from being infected. We now know that there was never any evidence to back this up. And when official data started to suggest that the vaccinated may be getting infected at similar if not greater rates than the unvaccinated, the response of some journalists was not to probe further and investigate but to encourage the authorities to suppress the data. Truly, you could not make it up.
Researchers such as Alex de Figueiredo from the London School of Hygiene and Tropical Medicine warned at the time that coercion was likely to lead to a loss of trust in vaccination more generally. With the latest research paper, we have firm evidence that this is indeed the case.
It is not unreasonable to conclude that the most dangerous ‘anti-vaxxers’ over the past two years have not been fringe conspiracy theorists but governments and the public health establishment itself.
It’s a sorry state of affairs, but where do we go from here? Well public health leaders could start by switching tack. Rather than setting out to persuade everyone to get vaccinated, they could concentrate on providing good information about benefits and risks to help people decide whether vaccination is right for them. If, at the same time, they start to engage honestly with those who have suffered side effects and acknowledge the high level of uncertainty that still exists in the evidence, perhaps they can begin to regain some of the trust they have so negligently lost.
David Paton is Professor of Industrial Economics at Nottingham University Business School.
Pfizer Press Release on New Bivalent Booster Raises More Questions Than It Answers
By Madhava Setty, M.D. | The Defender | November 9, 2022
Pfizer last week announced what some media outlets called “good news” about its COVID-19 bivalent booster, for which the U.S. Food and Drug Administration (FDA) in August granted Emergency Use Authorization on the basis of testing conducted on eight mice.
In a press release, Pfizer summarized the updated data from its phase 2/3 clinical trial on the Pfizer-BioNTech Omicron BA.4/BA.5-adapted bivalent COVID-19 vaccine:
- Bivalent booster elicited approximately 4-fold higher neutralizing antibody titers against Omicron BA.4/BA.5 sublineages compared to the original COVID-19 vaccine in individuals older than 55 years of age.
- One month after a 30-µg booster dose of the bivalent vaccine, Omicron BA.4/BA.5-neutralizing antibody titers increased 13.2-fold from pre-booster levels in adults older than 55 years of age and 9.5-fold in adults 18 to 55 years of age, compared to a 2.9-fold increase in adults older than 55 years of age who received the original booster vaccine.
- Safety and tolerability profile of bivalent booster remains favorable and similar to the original COVID-19 vaccine.
In simplest terms, the bivalent booster increased antibodies to the Omicron sublineages by a factor of 13.2, whereas the original booster increased them by a factor of only 2.9.
This increase was seen only in people over age 55.
Although the new booster produced a more modest increase (9.5 fold) in antibodies in the younger age group (18-55), Pfizer chose not to report what the response was in the age-matched group who received the original booster.
Did the bivalent booster offer any advantage in people younger than 55? Pfizer’s press release sidesteps this important question by focusing only on the results in the 55 or older cohort.
Dr. Ugur Sahin, CEO and co-founder of BioNTech, stated:
“These data demonstrate that our BA.4/BA.5-adapted bivalent vaccine works as conceptually planned in providing stronger protection against the Omicron BA.4 and BA.5 sublineages.”
By “these data” Sahin is referring only to those 55 or older as no comparative results were given in the 18-55 group. But how does he know that this rise in antibody levels will provide “stronger protection against the Omicron BA.4 and BA.5 sublineages?”
Were there any clinical outcome differences in terms of COVID-19 infections? None were recorded. Neither were any recorded in the preliminary data from the same study reported in a previous Pfizer press release.
This was likely why Sahin cautiously stated at that time:
“These preliminary findings are consistent with our preclinical data showing a substantial increase in the neutralizing antibody response against the Omicron sublineages BA.4 and BA.5.”
At what point does an “increase in the neutralizing antibody response” confer “stronger protection?”
Sahin doesn’t know — and neither does the FDA, which insisted that despite this uncertainty, the agency had enough grounds to cajole/compel/coerce those who acquired SARS-CoV-2 antibodies from a previous bout with COVID-19 to get jabbed anyway.
Questions about the two groups
The 36 people over age 55 who received the new bivalent formulation used in this comparison were taken from a larger pool of participants. They were “evenly stratified between those who had evidence of prior SARS-CoV-2 infection and those who did not.”
Furthermore, Pfizer informed us that the “control” group — those people who received the original booster formulation — were also a subgroup of participants but were taken from a different study “while ensuring the same equal stratification.” This group is thus called a “comparator group” and is not a true control group.
How did Pfizer decide which participants to use from the two separate studies?
Pfizer assured us the trial participants they used in the comparative group were “randomly selected.” However, we are not told how Pfizer selected those in the bivalent booster cohort.
Did they choose participants who had particularly high antibody responses? Were those in the comparative group actually chosen randomly — or were they chosen because of their particularly low responses?
In fairness, we are dissecting a press release and not any published research. However, given the company’s poor track record on transparency and integrity, these questions are not entirely unfounded.
In any case, there would be little incentive for Pfizer to cherry-pick participant data to showcase the benefit of its new bivalent booster. The Centers for Disease Control and Prevention (CDC) has already granted Pfizer Emergency Use Authorization for the bivalent booster.
However, Pfizer admitted that there was a difference between the two groups. The interval between the booster and the trial participant’s last exposure to a vaccine was significantly longer in those who received the bivalent booster (10-11 vs. 7 months). Could this difference affect the antibody responses in the two groups?
Pfizer shrugs this off, explaining that “Despite this difference, pre-booster antibody titers were similar for both.”
But pre-booster antibody titers were not the metric of interest — it was the difference in antibody titers before and after that Pfizer was seeking to compare. At least one study indicates that a longer interval between vaccine exposure results in a greater antibody response.
Furthermore, Pfizer’s results indicate that the antibody response was lower in those who had a previous bout of COVID-19, or as they put it, “greater in those without prior infection.”
As early as February 2022, the CDC reported that 57.7% of the U.S. population had been exposed to SARS-CoV-2 through seroprevalence studies. Not only is this most recently available data nine months old, but the CDC also admitted that this was an underestimate because breakthrough infections result in lower anti-N titers (vaccinated people who succumb to COVID-19 generate lower levels of anti-nucleocapsid antibodies than those who are unvaccinated).
In other words, unless you are in the minority and have not had COVID-19, you can look forward to a lower antibody response from the bivalent booster.
What about safety?
“The safety profile remains favorable for the bivalent vaccine and consistent with the original vaccine,” according to Pfizer.
And Pfizer knows this because three dozen bivalent booster recipients didn’t suffer any serious adverse events after one month of observation.
Apparently, Pfizer continues to believe that its original COVID-19 vaccine has a favorable safety profile — despite the more than 31,000 reports of deaths submitted to the Vaccine Adverse Event Reporting System, or VAERS (most occurring in the first seven days after inoculation), the 7.8% of vaccinated people who reported to V-safe that they sought medical attention after a COVID-19 vaccine and the nearly 80,000 serious adverse events and 1,223 deaths reported within three months of the vaccine’s roll-out, according to Pfizer’s own post-marketing analysis.
Ongoing studies will not determine any benefit
Despite reporting only inconsequential data in three dozen people over the age of 55, Pfizer announced in the same press release:
“A booster dose of the BA.4/BA.5-adapted bivalent vaccine has been authorized for emergency use by the FDA for ages 5 years and older and has also been granted marketing authorization in the EU by the European Commission following a positive opinion from the EMA for ages 12 years and older. An application for marketing authorization of the BA.4/BA.5 booster has been submitted to the EMA for children ages 5 through 11.”
And more good news:
“[Pfizer and BioNTech] also initiated a Phase 1/2/3 trial in September 2022, to evaluate the safety, tolerability and immunogenicity of different doses and dosing regimens of the companies’ Omicron BA.4/BA.5-adapted bivalent vaccine among children 6 months through 11 years of age.”
Yes. It is important for parents to know what dose their child can tolerate.
However, conspicuously missing in the design of this ongoing pediatric trial is the determination of efficacy — how good the product is in protecting children from contracting COVID-19 or from negative COVID-19 outcomes (death, hospitalization, ER visits, etc.). Isn’t that the point of a “vaccine” trial?
Apparently not — at least not according to the study description:
“The purpose of this clinical trial is to learn about the safety, extent of the side effects, and immune responses of the study vaccine (called bivalent BNT162b2 Omicron containing vaccine) in healthy children.”
Although having an estimated enrollment of 2,270 children, this ongoing trial will not answer the primary question parents will ask: Will this therapy benefit my kid?
Rather it will only allow Pfizer and BioNTech to determine how powerful their experimental mRNA product is in provoking infants and children to generate Omicron-specific antibodies.
Why are Pfizer and BioNTech not interested in proving their product does anything meaningful? Could it be because they realize this would be an impossible task?
Using data from nine months ago, the CDC reported that at least 75% of children in this age group had already acquired natural immunity to this disease. It is highly improbable that this study could demonstrate any differences in COVID-19 infection rates or outcomes, even with more than 2,000 children enrolled.
Madhava Setty, M.D. is senior science editor for The Defender.
This article was originally published by The Defender — Children’s Health Defense’s News & Views Website under Creative Commons license CC BY-NC-ND 4.0. Please consider subscribing to The Defender or donating to Children’s Health Defense.
Savior Al Gore wields windmills to stop the storms and end the Culture of Death

By Jo Nova | November 8, 2022
Like a conglomerate witch-doctor cum pagan-preacher the only thing Al Gore recycles is the overproof grade hellfire of centuries past. Chieftain Al will stop the storms if only everyone will do as he says and invest in his climate asset fund. For he cometh armed with windmills that stop rain-bombs and solar panels that hold back the sea.
Like medieval Occult leaders, superstitious rain dancers, and healers with magical cures, the modern witchdoctors have satellites and simulations, but run on the same old formula since time began. Fear, smear, demons, and magic. All prophesies are ambiguous. Nothing he sayth can be falsified.
Coal, apparently, is not just a source of emissions, but a veritable “culture of death.” Despite the era of coal being a time of record crop yields, bountiful food, travel, and exponential world population growth. Despite the working class of today being richer than the kings of centuries past.
One day, he promises, after he is safely dead, all weather will be good weather, and only the perfect amount of rain will fall, and ski seasons will start and end on the same day each year. Trust me, he says.
Damian Carrington, at The Guardian, swallows the whole prospectus:
Al Gore has pleaded for leaders to end what he calls ‘this culture of death’
The former US vice-president Al Gore is on fist-waving, passionate form in front of world leaders at Cop27. “We continue to use the thin blue atmosphere as an open sewer,” he says. “It is getting steadily worse. We have a credibility problem – all of us – we are not doing enough.”
Gore says we can continue the “culture of death” by continuing to dig up fossil fuels, and cites vast floods in Pakistan, heatwaves and “rain bombs” in China, and a million displaced in Nigeria.
And the valley of death will spread, and 1 billion immigrants will move into your home. (And you thought the southern US border crisis was bad?)
“The current areas of the world considered uninhabitable by doctors are small today but due to expand,” he says, with 1 billion migrants potentially crossing international borders this century, with all the colossal difficulties that would bring. If we stop subsidizing the culture of death and back renewable energy instead we can survive, Gore says, and no new fossil fuel projects are acceptable.
Here me O’ Minions — take this essence of arsenic, I mean lithium, and it shall cure your poverty:
Instead, Gore says, “Africa can be a renewable energy superpower”. He says 40% of world potential is in Africa.
Sure, Africa can be a renewable energy superpower, just like nowhere on Earth is. Europe can’t manage it, but Chad will show the way.
Of course, those who question their Occult Leader are racist pigs:
He says the dash for gas in Africa, a contentious issue at COP27, is a new form of colonisation, with the fuel to be sent to rich nations. He quotes the late archbishop Desmond Tutu as saying climate change is the apartheid of our time.
Somehow drilling gas in Africa for Europe is “colonisation” but using African children to mine cobalt for our batteries is the work of saints. Often the same saints want to segregate nations according to skin color. When did Al Gore say “All Lives Matter?” I can’t recall…
Toss in an attempt at being a Concern-Troll:
[Gore] also warns about stranded assets of billions, especially in Africa, if climate action closes oil and gas plants early.
Shucks. The last thing Al Gore wants is Africans to be lifted out of energy poverty.
So much better to keep casting those spells and doing rain dances. Africans are just his stage props.

