Tennessee Says ‘No’ to Vaccinating Children Without Parental Consent
By Brenda Baletti, Ph.D. | The Defender | April 21 , 2023
The Tennessee Legislature on Thursday passed a law that prevents healthcare providers in the state from vaccinating minors without parental consent. The bill is now awaiting Gov. Bill Lee’s signature.
The “Mature Minor Doctrine Clarification Act,” passed in the Senate last week and in the House Thursday, requires healthcare providers to obtain informed consent from a parent or legal guardian before vaccinating a minor.
During its COVID-19 vaccination campaign in 2021, the Tennessee Department of Health (DOH) invoked the “‘mature minor’ doctrine” — which says children of different ages have different maturities and capacities to consent to medical treatment.
The DOH used this doctrine to justify allowing minors ages 14 and older to consent to vaccination without informing their parents, as long as the child is deemed “mature” by their physician.
But Rolf Hazlehurst, senior staff attorney for Children’s Health Defense (CHD), said the DOH’s vaccination policy was based on a “dangerous oversimplification” of the mature minor doctrine.
Many Tennessee lawmakers agreed. During an April 10 subcommittee meeting, bill sponsor Rep. John Ragan (R-Oak Ridge) said the new law, then a bill, clarified the mature minor doctrine, which he said, “has been misinterpreted and shall we say, abused somewhat.”
Ragan added that by passing this law, legislators would be “giving parents back their rights to make medical decisions for their children.”
Hazelhurst told The Defender the law is “extremely important because it closes a legal loophole, which endangered parents, children and healthcare providers.”
In a Feb. 21 hearing of the Tennessee House Health Subcommittee, Hazelhurst testified that the law’s importance goes beyond Tennessee, because “nationwide, people are still relying upon Tennessee’s misinterpretation of the mature minor doctrine” to vaccinate children without parental consent.
Hazlehurst was the senior attorney in a lawsuit brought by CHD against the District of Columbia, seeking an injunction to block enforcement of a law passed in 2020 allowing children 11 and older to consent to vaccination.
In that case, Hazlehurst said, the district based the arguments in its briefs in part on the Tennessee DOH’s interpretation of the mature minor doctrine as legal precedent for its law.
The federal judge in the district case sided with the plaintiffs and issued an injunction against the law in March 2022.
The “mature minor” doctrine
The “mature minor” doctrine invoked by DOH is based on a 1987 Tennessee Supreme Court case, Cardwell v. Bechtol, where the court ruled in favor of a doctor accused of medical malpractice for treating a 17-year-old girl for a herniated disk.
In that case, the court said the common law “Rule of Sevens” established that children have different capacities to consent to different medical treatments as they grow up, differentiating among children under the age of 7, ages 7 to 14 and over 14.
The Tennessee DOH summarized the “mature minor” doctrine in a memo on its website, interpreting it to mean that children under age 7 cannot consent to treatment. For children between the ages of 7 and 14, “a physician generally should get parental consent before treating.”
And for children ages 14 to 17, “The physician may treat without parental consent unless the physician believes that the minor is not sufficiently mature to make his or her own health care decisions,” it says.
In all cases, there are certain statutory exemptions where physicians can treat children without parental consent, including in situations involving emergencies, drug abuse, and contraception and prenatal care.
Based on this summary of the doctrine, the DOH concluded that county health departments follow Tennessee law and “provide medical treatment and vaccinations to patients as young as 14 without parental consent if the individual provider determines that the patient meets the definition of a ‘mature minor.’”
But the DOH’s summary of the doctrine “omit[s] critical elements of the mature minor doctrine,” CHD argued in a letter sent in August 2021 to Ragan and Sen. Kerry Roberts.
The Cardwell v. Becthol ruling explicitly stated that the “adoption of the mature minor exception to the common law rule [used in the ruling] is by no means a general license to treat minors without parental consent.”
Rather, the application of the mature minor doctrine is dependent “on the facts of each case,” to be determined by a jury. In other words, the DOH was taking a jury instruction in a medical malpractice case and stating it was a statewide standard for vaccination, contrary to the spirit of the ruling, the CHD letter said.
“We do not, however, alter the general rule requiring parental consent for the medical treatment of minors,” the Tennessee Supreme Court ruling states.
History of the controversy
The Tennessee DOH fired Dr. Michelle Fiscus, medical director of Tennessee’s Vaccine-Preventable Diseases and Immunization Program, in the summer of 2021, in part in response to an email she sent on May 12, 2021.
Fiscus sent the email to COVID-19 vaccine providers across Tennessee informing them that according to Tennessee’s “mature minor” doctrine, they could provide vaccinations to minors as young as 14 without parental consent if they deemed that patient to be a “mature minor.”
The email stated: “There is no federal, legal requirement for parent or caregiver consent for COVID-19, or any other, vaccine.”
Fiscus sent the email the same day the Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices voted to recommend the use of Pfizer’s COVID-19 vaccines for children ages 12 and up, a decision she references in the email.
A few weeks after the letter went out, in the Tennessee General Assembly’s Joint Government Operations Committee meeting on June 16, 2021, Dr. Lisa Piercey, health commissioner and Fiscus’ boss, came under fire by Republican legislators for the email and the DOH’s targeting of children more generally.
Piercey confirmed in her testimony that based on the “mature minor” doctrine, the state of Tennessee treated 14-year-old children as capable of making decisions themselves as to whether to get the COVID-19 vaccine that was under Emergency Use Authorization.
Sen. Mark Pody responded, “I don’t know the terms that I could use to express my extreme disappointment, that in the state of Tennessee, where the majority of adults said no, to think that a 14-year-old child could say yes.”
At that time, only 37-39% of Tennessee residents were vaccinated, according to Piercey.
At the June 16, 2021, meeting, Rolf testified that the memo was clearly written by a lawyer — which Fiscus later confirmed in defending the email — in order to set a statewide locality rule that children 14 and up could be vaccinated without their parents’ consent.
“If a parent brings a lawsuit based on lack of consent, I have no doubt whatsoever that that letter will be attached as an exhibit in the motion to dismiss and the motion for summary judgment,” he said, adding, “I can’t stress enough that this letter has tremendous legal effect.”
The DOH fired Fiscus on July 12, 2021.
The Mature Minor Doctrine Clarification Act, which clarifies the rules about minor consent with respect to vaccination, was filed in the Tennessee General Assembly on Jan. 31.
‘Minor consent’ laws failing across the country
In March 2022, the U.S. District Court for the District of Columbia issued a preliminary injunction prohibiting the mayor of the District of Columbia, the D.C. Department of Health and D.C. public schools from enforcing the D.C. Minor Consent for Vaccinations Amendment Act of 2020 (D.C. Minor Consent Act), The Defender reported.
That law allowed a minor, 11 or older, to receive a vaccine, “if the minor is capable of meeting the informed consent standard” and the vaccine is recommended by the CDC’s Advisory Committee on Immunization Practices and provided according to the childhood immunization schedule.
The injunction stemmed from two lawsuits filed against the D.C. Minor Consent Act.
CHD and the Parental Rights Foundation filed one lawsuit, and the Informed Consent Action Network filed the other.
During oral arguments in the case, Hazlehurst argued the D.C. Minor Consent Act violates the Supremacy Clause of the U.S. Constitution because it contains multiple provisions that strip away the meager protections guaranteed to parents under the National Childhood Vaccine Injury Act of 1986.
The preliminary injunction, which the defendants did not appeal within the required 30-day period and therefore remains in place, reverted the district to the standard age of consent of 18, at least until the conclusion of the case.
Brenda Baletti Ph.D. is a reporter for The Defender. She wrote and taught about capitalism and politics for 10 years in the writing program at Duke University. She holds a Ph.D. in human geography from the University of North Carolina at Chapel Hill and a master’s from the University of Texas at Austin.
This article was originally published by The Defender — Children’s Health Defense’s News & Views Website under Creative Commons license CC BY-NC-ND 4.0. Please consider subscribing to The Defender or donating to Children’s Health Defense.
Facebook Should Keep Removing COVID ‘Misinformation,’ Oversight Board Says
By Brenda Baletti, Ph.D. | The Defender | April 21, 2023
The oversight board for Facebook’s parent company, Meta, on Thursday recommended the social media giant “maintain its current policy” of removing COVID-19 “misinformation” from its platform until the World Health Organization declares an end to the global pandemic.
The board made the recommendation despite widespread outcry about social media censorship after the Twitter Files and several ongoing lawsuits revealed collusion between state actors and social media companies to censor dissenting opinions and factual information that contradict official narratives, including those related to the COVID-19 pandemic.
The recommendation came in response to a request by Meta in July that the oversight board — an independent panel of tech and legal experts selected by Meta to weigh in on content policy issues — assess whether “a less restrictive approach” to censoring misinformation might “better align with its values and human rights responsibilities.”
Meta’s current misinformation policy sets different categories of harm content might cause, making that content subject to removal. Content is censored if the platform deems that it contributes to the “risk of imminent physical harm,” could cause “interference” with the functioning of political processes or contains “certain highly deceptive manipulated media.”
But the board didn’t find inconsistency between Meta’s “misinformation policy” and its “values and human rights responsibilities.” Instead, it said Meta’s current “exceptional measures” of eliminating disinformation are “justified.”
The board also urged Meta to “begin a process” to reassess which “misleading claims” it removes, to be more transparent about government requests for information, to consider making its “misinformation” policies more localized and to investigate how the architecture of the platform facilitates the spread of misinformation.
Meta said Thursday it will publicly respond to the board’s non-binding recommendations within 60 days.
Suzanne Nossel, a board member and CEO of PEN America, told The Washington Post that the board’s recommendations are not just relevant to COVID-19, but could shape Meta’s approach to anticipated future global health emergencies.
“The decision is less perhaps about the COVID pandemic per se or exclusively than about … how Meta should handle its responsibilities in the context of a fast-moving public health emergency,” she said.
How Facebook and Instagram censor COVID ‘misinformation’
The recommendation specifically assessed Meta’s “misinformation about health during public emergencies” policy, under which it removes 80 distinct “COVID-19 misinformation claims” posted on its platforms, such as claiming masking or social distancing lack efficacy or that the vaccines can have serious side effects.
Between March 2020 and July 2022, Facebook and Instagram, also owned by Meta, removed 27 million instances of COVID-19 “misinformation,” 1.3 million of which were restored on appeal.
The social media giant also designates a second type of COVID-19 “misinformation,” which does not reach the standard of removal, but is still subject to manipulation by the platform.
For example, information in that category is “fact-checked” where it is labeled as “false” or “missing context,” and then linked to a fact-checking article. That content is then also demoted so that it appears less frequently and prominently in users’ feeds.
Meta also treated other information with what it calls “neutral labels,” where it labeled posts with statements such as “some unapproved COVID-19 treatments may cause serious harm” and then directed people to Meta’s COVID-19 information center, which provides approved information from public health authorities.
Last July, the company said it had connected more than 2 billion people across 189 countries to “trustworthy information” through the portal. But it decided to stop using the neutral labels in December 2022, to ensure they would remain effective in other health emergencies, according to the oversight board’s report.
The basis for determining what is misinformation is whether the information conforms to what public health authorities deem to be true, according to the board’s recommendation and the Facebook policy page.
But throughout the pandemic, public health authorities have had to concede they were wrong about things — and that they lied about things — they had previously pronounced to be science-backed facts.
These “facts” include, for example, flip-flopping on masks, the lab-leak hypothesis, the effectiveness of natural immunity and numerous claims about vaccine efficacy, including that it stops transmission.
That means the platforms eliminated and demoted facts and information that were true. Even CNN conceded that “the company applied the labels to a wide range of claims both true and untrue about vaccines, treatments and other topics related to the virus.”
‘This kind of abuse of power should terrify all of us’
The board recommendations don’t mention the events that led Meta to consider changing its policies — controversy over recent revelations about how government officials coerced social media companies into toeing the government line.
In 2021, President Biden directly criticized Facebook and other platforms, saying they allowed “vaccine misinformation” to spread and they contributed to deaths from COVID-19.
He said they were “killing people” and that the pandemic was only “among the unvaccinated.”
Biden’s accusation was accompanied by threats of regulatory action from from high-ranking members of the administration — including White House Press Secretary Jennifer Psaki, Surgeon General Dr. Vivek Murthy and Department of Homeland Security (DHS) Secretary Alejandro Mayorkas — if the social media companies did not comply.
Psaki said government officials were in regular touch with social media platforms, telling them what — and in some cases whom — to censor, Jenin Younes reported.
DHS even created a video in 2021, since removed from youtube, encouraging children to report their own family members to Facebook for ‘disinformation’ if they challenge U.S. government narratives on COVID-19.
Writing in Tablet Magazine this month, civil liberties attorney Jenin Younes recounted the story of a Facebook support group for people who experienced adverse events related to the COVID-19 vaccines being shut down for spreading harmful “misinformation.”
Last month, in the Twitter Files release about Stanford University’s Virality project, Matt Taiibbi revealed that Stanford, with the backing of several government agencies, had created a cross-platform digital ticketing system that was processing censorship requests for all of the social media platforms, including Meta’s.
The Virality Project claimed its objective “is to detect, analyze, and respond to incidents of false and misleading narratives related to COVID-19 vaccines across online ecosystems.”
Taibbi said the Virality Project was “defining true things as disinformation or misinformation or malformation,” which he said signifies “a new evolution of the disinformation process away from trying to figure out what’s true and what’s not and just going directly to political narrative.”
That reflects Meta’s policy to censor statements that don’t conform to official public health authority doctrine as “misinformation.”
Meta’s policies do not mention the tips and directions it receives from government agencies about misinformation.
Sen. Rand Paul (R-Ky.) on Tuesday published an op-ed in The Hill calling for an end to censorship practices, pointing out that statements about COVID-19 made on platforms like Facebook that are now supported by evidence were flagged as disinformation.
”Statements including my own, that our government once labeled as ‘disinformation,’ such as the efficacy of masks, naturally acquired immunity, and the origins of COVID-19, are now supported by evidence,” he said.
“In reality, the most significant source of disinformation during the pandemic, with the most influence and greatest impact on people’s lives, was the U.S. government,” he added.
Rand pointed to critiques of DHS’s “abusive practices” by organizations like the American Civil Liberties Union and highlighted a Brennan Center for Justice report published last month that found at least 12 DHS programs for tracking what Americans are saying online.
“This kind of abuse of power should terrify all of us regardless of which side of the aisle you are on,” he said.
Brenda Baletti Ph.D. is a reporter for The Defender. She wrote and taught about capitalism and politics for 10 years in the writing program at Duke University. She holds a Ph.D. in human geography from the University of North Carolina at Chapel Hill and a master’s from the University of Texas at Austin.
This article was originally published by The Defender — Children’s Health Defense’s News & Views Website under Creative Commons license CC BY-NC-ND 4.0. Please consider subscribing to The Defender or donating to Children’s Health Defense.
CDC director gives misleading testimony to Congress
Walensky misled Congress on vaccine effectiveness against viral transmission and on Cochrane review of face masks
BY MARYANNE DEMASI, PHD | APRIL 20, 2023
This week, CDC director Rochelle Walensky provided witness testimony to the House Committee on Appropriations responsible for overseeing the funding of various federal programs related to labour, health, education, and other related agencies.
But serious questions have been raised about the veracity of Walensky’s testimony.
Congressman Andrew Clyde (R-Ga) asked Walensky if her March 2021 public statement on MSNBC, in which she unequivocally said that “vaccinated people do not carry the virus, they do not get sick” was accurate.
“At the time it was [accurate]” Walensky replied confidently.
She then proceeded to explain, “We’ve had an evolution of the science and an evolution of the virus” and that “all the data at the time suggested that vaccinated people, even if they got sick, could not transmit the virus.”
However, there was no such evidence at the time and it prompted criticism from scientists who said there weren’t enough data to claim that vaccinated people were completely protected or that they could not transmit the virus to others.
One of those critics was Jay Bhattacharya, professor of health policy at Stanford University School of Medicine.
“Back then, Walensky didn’t know if it was true. It was just an irresponsible use of a bully pulpit as a CDC director to say something that she did not know for certain to be true at the time,” said Bhattacharya.
“Unfortunately, people used that information to discriminate against unvaccinated individuals and would certainly have been used as fuel for very destructive policies like vaccine mandates,” he added.
Notably, only days after Walensky made that statement to MSNBC, a spokesperson from her own agency had to walk back the comments saying, “Dr Walensky spoke broadly in this interview” adding that it was possible for fully vaccinated people to get COVID-19.
Walensky missed the memo
Walensky should have known that when mRNA vaccines were first authorised in 2020, the FDA listed critical ‘gaps’ in the knowledge base. One of them was the vaccine’s unknown effectiveness against viral transmission.
Also, in Pfizer’s and Moderna’s original pivotal trials, there were 8 and 11 people respectively, who developed symptomatic COVID-19 in the vaccine group, proving the vaccines never had absolute effectiveness, like Walensky had claimed.

Several months later, the FDA’s evaluation stayed the same. In a clinical review, the FDA wrote, “remaining uncertainties regarding the clinical benefits of BNT162b2 in individuals 16 years and older, include its level of protection against asymptomatic infection and transmission of SARS-CoV-2, including for the delta variant.”
Even today, the FDA remains clear that efficacy against transmission is unproven. The FDA’s website states, “While it is hoped this will be the case, the scientific community does not yet know if Comirnaty will reduce such transmission.”
Walensky says Cochrane summary ‘retracted’
Another astonishing falsehood made by Walensky was her response to Congressman Clyde’s question about the Cochrane review which found that wearing face masks in the community “probably makes little to no difference” in preventing viral transmission.
Walensky enthusiastically stated, “I think its notable, that the Editor-in-Chief of Cochrane, actually said that the summary of that review was…[stumble]..she retracted the summary of that review and said that it was inaccurate.”
However, the summary of the review was not retracted, nor have the authors of the review changed the language in the summary.
Misleading statements by New York Times columnist Zeynep Tufekci has likely led to this falsehood being repeated (which I cover in a previous article).
In response to Walensky’s comments, Tom Jefferson, lead author of the Cochrane study said, “Walensky is plain wrong. There has been no retraction of anything.”
“It’s worth reiterating that we are the copyright holders of the review, so we decide what goes in or out of the review and we will not change our review on the basis of what the media wants or what Walensky says,” remarked Jefferson.
Bhattacharya was also stunned by Walensky’s comments. “It’s irresponsible for her to claim that the Cochrane review [summary] was retracted when it was not. It damages her credibility and harms the scientific process, which requires public officials to be honest about scientific results,” he said.
Did Walensky lie to Congress or is she poorly informed?
Witnesses at these hearings are expected to provide truthful and accurate information to the committee and may be subject to legal penalties if they provide false information or knowingly make false statements.
But will Walensky be held accountable for misleading Congress? Unlikely.
The Novelty of mRNA Viral Vaccines and Potential Harms: A Scoping Review
By Peter A. McCullough, MD, MPH | Courageous Discourse | April 19, 2023
We all have the tendency to paint issues with a broad brush. That is to see things one way for intellectual simplicity. “All pharmaceuticals are bad” or “I don’t trust any vaccine.” It is even more tempting to take a negative view on all new technology when the product launch in humans fails to a large degree.
These old mental saws could apply to mRNA vaccines. Halma et al have published a scoping review of lipid nanoparticle-mRNA products with fair balance causing the reader to consider future possibilities. The COVID-19 vaccines are known to be unsafe for several reasons: 1) the Wuhan Spike protein damages cells, tissues, organs, and causes blood clotting, 2) the lipid nanoparticles may have toxicity from the PEG or polysorbate 80 or from syncytia formation, 3) the mRNA appears to be resistant to ribonucleases and is not broken down in the body. As some point the mRNA or fragments could interfere with gene function or alter other microRNAs that are managing the human genome.

Halma, M.T.J.; Rose, J.; Lawrie, T. The Novelty of mRNA Viral Vaccines and Potential Harms: A Scoping Review. J 2023, 6, 220-235. https://doi.org/10.3390/j6020017
The Halma paper points out that safe mRNA products are possible. For example, properly designed mRNA coding for normal proteins that are deficient or ones that are sufficiently humanized and not recognized by the body as foreign could indeed become part of the future pharmacopeia. But there is no doubt that the first use of mRNA on a mass, indiscriminate scale has been a disaster with the COVID-19 vaccine campaign.
The Emergency is Dead – Long Live the PREP Act
The Naked Emperor’s Newsletter | April 19, 2023
Did you think the Covid emergency was over? President Biden certainly did.

But the Public Readiness and Emergency Preparedness (PREP) Act didn’t get the memo. On Friday, Secretary Becerra at the US Department of Health & Human Services (HHS) announced that he intends to amend the declaration under the Act for medical countermeasures against COVID-19.
So instead of repealing the Act, parts of it are to be extended – never let a good crisis go to waste, even if it’s the end of the crisis.
The PREP Act declaration deals with vaccines, tests and treatments and provides flexibilities and protections to individuals and entities involved in providing them. This includes liability protections for entities engaged in manufacturing, distribution or administration of these Covid countermeasures.
The announcement says that even once vaccines, tests and treatments move away from being distributed under a US Government agreement and they transition to traditional pathways, the PREP Act still won’t automatically terminate.
So what will remain even after the “emergency” has finally ended?
- Coverage for Covid vaccines, seasonal influenza vaccines and Covid tests. Immunity from liability will be extended until December 2024 to pharmacists, pharmacy interns and pharmacy technicians to administer Covid and flu jabs (to over 3s) and Covid tests, regardless of any government agreement or emergency declaration;
- Federal agreements related to the provision of Covid countermeasures (including vaccines and treatments) will also be extended until December 2024;
- There will be no impact on government distributed Covid countermeasures;
- Coverage for prescribing and dispensing of Covid-19 oral antivirals will not change. This includes liability immunity for dispensing Covid treatments such as Paxlovid and Lagevrio; and
- There will be no change to the “test to treat” program.
They’ve managed to extend the crisis that has just ended to at least December 2024, more than another year and a half – bravo! And they’ve managed to sneak in flu vaccines as part of the emergency as well.
I just can’t think why they would have to extend liability immunity of these products for so long? (sarcasm).
Oh and unvaccinated non-citizens still won’t be able to travel to the US, even once the emergency is over.
Once the power-hungry politicians seize power they just can’t seem to let it go.
Doubled Pregnancy Loss Rate, Raised Foetal Abnormality Rate and Concentration of Lipid Nanoparticles in Ovaries – How Could They Call This Vaccine ‘Safe’?
BY ALEX KRIEL AND DR DAVID BELL | THE DAILY SCEPTIC | APRIL 14, 2023
The mRNA vaccines were released globally in early 2021 with the slogan ‘safe and effective’. Unusually for a new class of medicine, they were soon recommended by public health authorities for pregnant women. By late 2021, working age women, including those who were pregnant, were being thrown out of employment for not agreeing to be injected. Those who took the mRNA vaccines did so based on trust in health authorities – the assumption being that they would not have been approved if the evidence was not absolutely clear. The role of regulatory agencies was to protect the public and, therefore, if they were approved, the drugs were safe.
Recently, a lengthy vaccine evaluation report sponsored by Pfizer and submitted to the Australian regulator, the Therapeutic Goods Administration (TGA) dated January 2021 was released under a Freedom of Information request. The report contains significant new information that had been supressed by the TGA and by Pfizer itself. Much of this relates directly to the issues of safety in pregnancy and impacts on the fertility of women of child-bearing age. The whole report is important, but four key data points stand out:
- The rapid decline in antibody and T cells in monkeys following a second dose;
- Biodistribution studies (previously released in 2021 through an FOI request in Japan);
- Data on the impact of fertility outcomes for rats;
- Data on foetal abnormalities in rats.
We focus on the last three items as, for the first point, it is enough to quote the report itself: “Antibodies and T cells in monkeys declined quickly over five weeks after the second dose of [Pfizer Covid vaccine] BNT162b2 (V9), raising concerns over long term immunity.” This point indicates that the regulators should have anticipated the rapid decline in efficacy and must have known at the outset that the initial two dose course was unlikely to confer lasting immunity and would, therefore, require multiple repeat doses. This expectation of failure was recently highlighted by Dr. Anthony Fauci, former director at the U.S. NIH.
The three remaining items should be a major cause for concern with the pharmaceutical regulatory system. The first, as revealed in 2021, involved biodistribution studies of the lipid nanoparticle carrier in rats, using a luciferase enzyme to substitute for the mRNA vaccine. The study demonstrated that the vaccine will travel throughout the body after injection and is found not only at the injection site but in all organs tested, with high concentration in the ovaries, liver, adrenal glands and spleen. Authorities who assured vaccinated people in early 2021 that the vaccine stays in the arm were, as we have known for two years, not being honest.
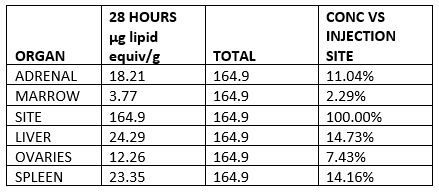
Lipid concentration per gram, recalculated as percentage of injection site.
In terms of the impact on fertility and foetal abnormalities, the report includes a study of 44 rats and describes two main metrics, the pre-implantation loss rate and the number of abnormalities per foetus (also expressed per litter). In both cases the metrics were significantly higher for vaccinated rats than for unvaccinated rats.
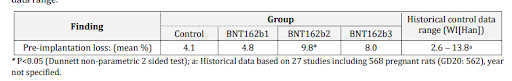
Roughly speaking, the pre-implantation loss ratio compares the estimated number of fertilised ova and the ova implanted in the uterus. The table below is taken from the report itself and clearly shows the loss rate for vaccinated (BNT162b2) is more than double the unvaccinated control group.

In a case control study, a doubling of pregnancy loss in the intervention group would represent a serious safety signal. Rather than take this seriously, the authors of the report then compared the outcomes to historical data on other rat populations – 27 studies of 568 rats – and ignored the outcome because other populations had recorded higher overall losses. This range is shown in the right hand column as 2.6% to 13.8%. This analysis is alarming as remaining below the highest previously recorded pregnancy loss levels in populations elsewhere is not a safe outcome when the intervention is also associated with double the harm of the control group.
A similar pattern is observed for foetal malformations with higher abnormality rate in each of the 12 categories studied. Of the 11 categories where Pfizer confirmed the data are correct, there are only two total abnormalities in the control group, versus 28 with the mRNA vaccine (BNT162b2). In the category which Pfizer labelled as unreliable (supernumerary lumbar ribs), there were three abnormalities in the control group and 12 in the vaccinated group.
As with the increased pregnancy losses, Pfizer simply ignored the trend and compared the results with historical data from other rat populations. This is very significant as it is seen across every malformation category. The case control nature of the study design is again ignored, in order to apparently hide the negative outcomes demonstrated.
These data indicate that there is no basis for saying the vaccine is safe in pregnancy. Concentration of lipid nanoparticles in ovaries, a doubled pregnancy loss rate and raised foetal abnormality rate across all measured categories indicates that designating a safe-in-pregnancy label (B1 category in Australia) was contrary to available evidence. The data imply that not only was the Government’s ‘safe and effective’ sloganeering not accurate, it was totally misleading with respect to the safety data available.
Despite the negative nature of these outcomes, the classification of this medicine as a ‘vaccine’ appears to have precluded further animal trials. Historically, new medicines, especially in classes never used in humans before, would require a very rigorous assessment. Vaccines, however, have a lower burden of proof requirement than ordinary medicines. By classifying mRNA injections as ‘vaccines’, this ensured regulatory approval with significantly less stringent safety requirements, as the TGA itself notes. In fact, these mRNA gene therapy products (to use the proper term) function more like medicines than vaccines in that they modify the internal functioning of cells, rather than stimulating an immune response to presence of an antigen. Labelling these gene therapy products as ‘vaccines’ means that, as far as we are aware, even today no genotoxicity or carcinogenicity studies have been carried out.
This report, which was only released after a FOI request, is extremely disturbing as it shows that authorities knew of major risks with mRNA COVID-19 vaccination while simultaneously assuring populations that it was safe. The fact that mainstream media have (as far as we are aware) completely ignored the newly released data should reinforce the need for caution when listening to the advice of public health messaging regarding COVID-19 vaccination.
Firstly, it is clear that regulators, drug companies and the Government would have known that vaccine induced immunity tails off very rapidly, with this being subsequently observed in real world data with efficacy against infection falling to zero. Accordingly, the single point in time figures of 95% and 62% efficacy against symptomatic PCR-positive infection quoted for Pfizer and AstraZeneca respectively meant almost nothing since a rapid decline was to be expected. Similarly, the concept of a two dose course was inaccurate as endless boosters would likely have been required given the rapid decline in antibodies and T-cells observed in the monkeys.
Most importantly, the data do not in any way support the ‘safe’ conclusion with respect to pregnancy; ‘dangerous’ would be much more accurate. The assurances of safety were, therefore, completely misleading given the data disclosures in the recent freedom of information release. Regulatory authorities knew that animal studies showed major red flags regarding both pregnancy loss and foetal abnormalities, consistent with the systemic distribution of the mRNA they had been hiding from the public. Even in March 2023, it is impossible to give these assurances, given the fact that important studies have not, to the best of our knowledge, been done. Pfizer elected not to follow up the vast majority of pregnancies in the original human trials, despite high miscarriage rates in the minority they did follow. Given all of the problems with efficacy and safety, the administration of these products to women of childbearing age and administration to healthy pregnant women is high risk and not justified.
Alex Kriel is by training a physicist and was one of the first people to highlight the flawed nature of the Imperial Covid model; he is a founder of the Thinking Coalition which comprises a group of citizens who are concerned about Government overreach. David Bell is a public health physician with a PhD in population health and formerly worked as a scientific and medical officer at the World Health Organisation.
U.S. Approves First 3 COVID Vaccine Injury Claims — And Pays Out a Total of $4,634.89
By Brenda Baletti, Ph.D. | The Defender | April 13, 2023
The U.S. government approved its first three payments to people injured by COVID-19 vaccines — amounting to a total of $4,634.89.
The Health and Resources Service Administration (HRSA) vaccine injury claims report, updated monthly, shows one $2,019.55 payment for anaphylaxis and two payments — $1,582.65 and $1032.69 — for myocarditis.
The payments were made through HRSA’s Countermeasures Injury Compensation Program (CICP).
The CICP was established under the Public Readiness and Emergency Preparedness (PREP) Act, which protects pharmaceutical companies from liability for injuries sustained from “countermeasures,” such as vaccines and medications, administered during a public health emergency.
Since 2010, when it approved its first claim, the program has compensated a total of 33 claims for vaccine injuries — but these are the first awards for COVID-19 vaccines.
“These long-awaited awards were overdue, highly anticipated and speculated upon,” said Kim Mack Rosenberg, acting general counsel for Children’s Health Defense (CHD). “What is remarkable is that less than $5,000 was paid — total. This is a tragedy that highlights the severe limitations of the program.”
CHD Acting President Laura Bono called the payouts for myocarditis “insulting,” given that mortality rates increase to 50% within five years of diagnosis.
Bono said:
“The CICP is a pathetic, government-run program that gives complete liability protection to the very industries profiting from the COVID vaccine or product. While victims linger with their injuries, paying out-of-pocket for expenses, or at worst die, the industries run to the bank.”
Since the start of the pandemic, people claiming injuries related to COVID-19 vaccines and other countermeasures submitted 11,425 requests for compensation.
Of those, only 19 have been declared eligible for compensation and are undergoing a “medical benefits review” to determine payment.
The anaphylaxis case had been pending medical benefits review since the fall of 2021, and the two myocarditis cases had been pending review since January.
During the medical benefits review, HRSA determines any costs remaining after insurance, workers’ comp, disability or other reimbursements or payments.
Wayne Rohde, an expert in vaccine injury compensation, wrote on his Substack that given the “18+ months to review previous medical benefits that may have been awarded to the injured party [the anaphylaxis case], this process tells me it was a major injury that resulted in very large medical bills.”
Myocarditis is a serious condition that also requires a lot of medical attention, Rohde said.
To date, there have been 1,541,275 reports of adverse events following COVID-19 vaccination submitted to the Vaccine Adverse Event Reporting System (VAERS).
How does vaccine injury compensation work? the VICP and CICP
HRSA, which operates under the U.S. Department of Health and Human Services (HHS), administers two vaccine injury compensation programs: the National Vaccine Injury Compensation Program (VICP) and the Countermeasures Injury Compensation Program (CICP).
The VICP is a special, no-fault tribunal housed within the U.S. Court of Federal Claims that handles injury claims for 16 common vaccines on the childhood vaccination schedule. To date, it has awarded more than $4 billion for medical bills, lost wages, lawyer fees, and pain and suffering to thousands of people for vaccine injuries.
The program does not currently cover COVID-19 vaccine injuries. Should COVID-19 vaccines be moved into the program, any injuries would be handled by the already overwhelmed VICP.
The CICP, the only program that covers COVID-19 vaccine injuries at this time, is even less equipped to deal with them, Rohde told The Defender.
“For COVID-19 vaccine-injured people, the CICP is the worst place, it’s the worst option,” Rohde said, “because it is not really a compensation program, it’s a reimbursement program for medical costs.”
The CICP allows individuals to claim compensation only for unreimbursed medical expenses — meaning those not fully reimbursed by insurance or government programs, such as Medicaid — for death and for lost wages up to $50,000.
According to the Centers for Disease Control and Prevention, under the CICP:
“Eligible individuals may be compensated for certain reasonable and necessary medical expenses and for lost employment income at the time of the injury. Death benefits may be paid to certain survivors of covered countermeasures recipients who have died as a direct result of the covered countermeasure injury.
“The U.S. Department of Health and Human Services is the payer of last resort. Therefore, payments are reduced by those of other third-party payers.”
“There’s no pain and suffering here, there’s nothing,” Rohde said.
Because the CICP reviews and resolves claims through an administrative rather than a judicial process, no details other than the amount of the payments have to be shared with the public.
“It’s designed to be very convoluted, very non-transparent,” Rohde said.
The CICP was known for its cumbersome claims process and low likelihood of success for claimants even before the pandemic. Since then, it has seen unsustainable growth.
According to HRSA’s numbers, of the 11,941 claims filed with the CICP since 2010, nearly 11,000 of them are still under review.
The HRSA budget for COVID-19 vaccine injury compensation will increase in fiscal year 2023 — from approximately $1 million to $5 million — and its budget for staffing and contractors will jump from $5 million to $9.5 million.
How would COVID vaccine injury compensation change under the VICP?
For vaccine injury claims to be covered under the VICP rather than the CICP, three requirements must be met:
- The vaccine needs to be recommended for routine administration for children and/or pregnant women.
- It needs to have an excise tax imposed upon it through the legislature.
- There needs to be a notice of coverage published in the federal record.
The COVID-19 vaccine was added to the childhood immunization schedule earlier this year — but the next two steps in the process have yet to be completed and public health officials have not indicated when this might happen.
The National Academies of Science, Engineering, and Medicine met for three days last month — behind closed doors, except for a two-hour public comment period — to review the epidemiological, clinical and biological evidence on adverse events associated with COVID-19 vaccines.
The committee will generate a report that will be used to add injuries to the federal Vaccine Injury Table, which lists known adverse events associated with existing vaccines.
This list helps the VICP and the CICP decide whether to compensate vaccine injury claims.
At the National Academies meeting, Professor Renee Gentry, director of the Vaccine Injury Litigation Clinic at The George Washington University Law School, told the committee the stakeholders that created the VICP — vaccine manufacturers, lawyers and parents — set it up to be petitioner-friendly, informal, generous and non-adversarial.
But instead, she said, HHS has been “unrelenting” in its opposition to recognizing vaccine injuries.
“I believe the VICP as it exists today would be unrecognizable to those original stakeholders,” she said.
Brenda Baletti Ph.D. is a reporter for The Defender. She wrote and taught about capitalism and politics for 10 years in the writing program at Duke University. She holds a Ph.D. in human geography from the University of North Carolina at Chapel Hill and a master’s from the University of Texas at Austin.
This article was originally published by The Defender — Children’s Health Defense’s News & Views Website under Creative Commons license CC BY-NC-ND 4.0. Please consider subscribing to The Defender or donating to Children’s Health Defense.