Does the Meningococcal Vaccine Used in the U.S. Stop Infection and Transmission of Meningococcal Bacteria?
Your bite-size dose of immunity against vaccine misinformation. Spread the truth.

Injecting Freedom by Aaron Siri | April 2, 2024
Does the meningococcal vaccine (MenACWY) used in the United States stop infection and transmission of meningococcal bacteria?
“Yes” or “No”?
When picking an answer, keep in mind that MenACWY is one of the vaccines mandated to attend secondary school in most U.S. states (and to attend university/college in many states), and the justification for these rights-crushing mandates is to prevent transmission of meningococcal bacteria in the school setting.
According to the CDC, “Rates of meningococcal disease have declined in the United States since the 1990s and remain low today. Much of the decline occurred before the routine use of MenACWY vaccines. … [D]ata suggest MenACWY vaccines have provided protection to those vaccinated, but probably not to the larger, unvaccinated community (population or herd immunity).”
Also consider that according to the CDC, “Protection from MenACWY vaccination wanes in most adolescents within 5 years,” and the CDC does not recommend routine administration of this vaccine for adults. And as for the claimed protection from this vaccine, its clinical trial did not prove it prevented a single case of meningococcal disease (relying instead on antibody levels, not clinical findings).
Screenshots of the relevant portions of the websites linked above (in case they change).
‘Shocking Cover-up’: DOJ Lawyers Committed Fraud in Vaccine Injury Case, CHD Attorney Alleges in Motion Filed Today
By Brenda Baletti, Ph.D. | The Defender | April 2, 2024
Rolf Hazlehurst, a Children’s Health Defense (CHD) staff attorney and father of a son with autism, today filed a motion in federal court alleging lawyers representing the U.S. Department of Health and Human Services (HHS) fraudulently concealed evidence that vaccines can cause autism.
In a motion filed in the U.S. Court of Federal Claims, Hazlehurst alleged that U.S. Department of Justice (DOJ) lawyers who represented HHS in vaccine injury cases repeatedly defrauded the judicial system — from the National Vaccine Injury Compensation Program (NVICP) to the U.S. Supreme Court.
That fraud led to thousands of families of vaccine-injured children being denied the right to compensation and the right to have their cases heard, according to the motion.
“This motion makes very serious and well-substantiated allegations of a massive scheme of fraud on the courts,” said Kim Mack Rosenberg, CHD general counsel who also is of counsel to Hazlehurst in the federal case.
“The evidence submitted in support of the motion clearly shows that attorneys from the Department of Justice concealed and misrepresented highly relevant information from the special masters in the Vaccine Injury Compensation Program and the judges in the courts,” Mack Rosenberg told The Defender.
Hazlehurst’s son Yates regressed into autism after being vaccinated as an infant. In the early 2000s, his family and thousands of others filed cases seeking compensation for vaccine-induced autism through the NVICP.
The program consolidated all of the petitions into the Omnibus Autism Proceeding (OAP) and selected six representative “test cases” — of which Yates’ was the second — as the basis for determining the outcome of the remaining 5,400 cases.
Unbeknownst at the time to the petitioners and the NVICP special masters, the DOJ’s star expert medical witness, Dr. Andrew Zimmerman informed DOJ attorneys during the ongoing omnibus proceedings that he had reversed his original opinion and determined that vaccines can and do cause autism in some cases.
In what Hazlehurst alleges was “a shocking cover-up,” instead of allowing Zimmerman to share his revised opinion, the DOJ attorneys relieved Zimmerman of his duties as a witness.
However, they continued to use excerpts from his unamended written opinion to make their case that vaccines did not cause autism — misrepresenting his position and committing “fraud on the court.”
According to the motion, the DOJ’s first act of fraud snowballed into a scheme of deception with far-reaching implications in which DOJ attorneys repeatedly misrepresented Zimmerman’s opinion and concealed other evidence that emerged during the test case hearings in the OAP in subsequent cases before multiple courts.
“As a result, thousands of cases in the Omnibus Autism Proceeding were denied compensation and the impact beyond the OAP is enormous,” Mack Rosenberg said. “This fraud affected the Vaccine Injury Compensation Program — especially the Omnibus Autism Proceeding — the Court of Federal Claims, the Court of Appeals for the Federal Circuit and even the U.S. Supreme Court.”
Hazlehurst said he is “asking the court to give this motion the serious attention it deserves.” He added, “At a minimum, the court should allow discovery and hold a hearing on this motion.”
Overturning a ruling due to fraud on the court is an extraordinary remedy reserved for extraordinary cases but according to Hazlehurst, “This motion we filed shows that this indeed is an extraordinary case.”
The DOJ has until April 30 to respond to the motion.
CHD CEO Mary Holland told The Defender, “Vaccines most definitely do cause autism, and the government has been lying about this reality for decades.”
Holland added:
“With others, I published a law review article in 2011 showing that the government absolutely knew that vaccines cause autism — and yet they have covered it up and lied about it since the inception of the Vaccine Injury Compensation Program.
“How many hundreds of thousands of children and families would have been spared the heartaches and crushing financial burdens of autism had the government come clean?”
‘Exceptionally difficult’ to obtain compensation through NVICP
In the late 1980s, a substantial number of lawsuits for vaccine injuries related to Wyeth’s (now Pfizer) DPT vaccine, combined with “grossly insufficient compensation” for victims of vaccine injury, threatened the vaccine program’s viability.
In response, Congress passed the National Childhood Vaccine Injury Act of 1986, which established the “vaccine court.” The law gave the pharmaceutical industry broad protection from liability and proposed to compensate vaccine-injured children through the new NVICP.
The NVICP originally was designed to be a “swift, flexible, and less adversarial alternative to the often costly and lengthy civil arena of traditional tort litigation.”
To receive compensation, parents file a claim with the program.
The Court of Federal Claims (which oversees the program) appoints “special masters” — typically lawyers who previously represented the U.S. government — to manage and decide the individual claims. Attorneys may represent the petitioners, and the DOJ represents HHS.
NVICP proceedings are more informal than a typical courtroom. Unlike regular court proceedings, petitioners in the “vaccine court” have no right to discovery.
If a petitioner files a claim for a vaccine covered under the program and listed on the Vaccine Injury Table — the list of known vaccine side effects associated with certain vaccines within set time frames — it is presumed that a vaccine caused the petitioner’s injury and the petitioner is eligible for compensation without proof of causation.
However, if a petitioner experiences an “off-table injury” — an injury not listed on the table or that didn’t happen in the recognized injury time frame — the petitioner must prove by “a preponderance of evidence” that the vaccine caused the injury. Evidence includes medical records and expert witness testimony.
Claims must be filed within three years of the first symptom or two years of death.
Petitioners must provide a medical theory of the cause, a sequence of cause and effect, and show a temporal relationship between vaccine and injury.
However, the NVICP does not specify the required volume and type of evidence, so meeting the “preponderance of evidence” standard is largely at the discretion of the special master.
Petitioners can appeal NVICP cases to the Court of Federal Claims, the Court of Appeals for the Federal Circuit and ultimately to the U.S. Supreme Court.
It is “exceptionally difficult” to obtain compensation within the NVICP, Hazlehurst told The Defender. The proceedings are often turned into drawn-out, contentious expert battles and the backlog of cases is substantial.
The Vaccine Act of 1986 is unjust for petitioners, Hazlehurst alleges. And that injustice reached its zenith with the OAP, when the DOJ perpetrated fraud right under the noses of the special masters, signaling the beginning of the fraud on the courts that continues to this day.
Hazlehurst told The Defender he hopes his motion will shed light on the damage inflicted by this law and that it will ultimately help end the autism epidemic.
“The Vaccine Act of 1986 is one of the fundamental causes of the autism epidemic,” Hazlehurst said. “Understanding why this is true, and how the United States Department of Justice perpetrated fraud upon the courts, including the Supreme Court of the United States, is the key to ending the autism epidemic.”
A short history of the autism omnibus proceedings
By 2002, to address a “massive influx” of petitions alleging vaccine-induced autism, the Office of Special Masters combined over 5,000 claims into the OAP to determine whether vaccines cause autism and if so, under what conditions.
Initially, the NVICP planned to investigate causation issues and apply those general findings to individual cases. However, the program changed its strategy and instead selected six “test cases” by which it would examine the evidence for injuries caused by the measles mumps rubella (MMR) vaccine, thimerosal-containing vaccines (TCV), or a combination of both.
Then it would apply the findings of the test cases to other similar cases.
In doing so, Hazlehurst alleges, the court conflated general causation evidence with specific causation evidence from a few cases, without allowing for rules of discovery or evidence that would apply in an actual court.
This, Hazlehurst said, “was a recipe for disaster” as each test case was then used to determine the outcome for the remaining 5,000 cases.
Three cases — Cedillo v. HHS, Hazlehurst v. HHS and Poling v. HHS — are at the center of the alleged fraud by the DOJ.
Fraud #1: the Zimmerman testimony
Hearings for the first OAP test case, Cedillo v. HHS, began in 2007. Zimmerman had worked with the DOJ to prepare an expert report on behalf of HHS finding that Michelle Cedillo’s autism had likely not been caused by the MMR vaccine.
Zimmerman later wrote in a 2018 affidavit that he attended the Cedillo hearing and listened to the testimony of Dr. Marcel Kinsbourne, another world-renowned expert in pediatric neurology.
On that basis, Zimmerman stated, he decided to clarify his written expert opinion about Michelle Cedillo, concerned it would be taken out of context.
Zimmerman spoke with DOJ attorneys to clarify that his expert opinion in the Cedillo case “was not intended to be a blanket statement as to all children and all medical science,” according to the 2018 affidavit.
He specified that advances in science, medicine and his own clinical research had led him to believe there were exceptions in which vaccinations could cause autism.
He also referred the attorneys to a paper he published with colleagues in 2006, the Poling paper, describing the case of an unidentified child who suffered regressive autism following vaccine adverse reactions. The paper suggested a possible association between mitochondrial dysfunction, vaccinations and regressive autism.
After communicating this evidence to DOJ attorneys, the DOJ dismissed Zimmerman as a witness but continued to use his written opinion as general causation evidence.
The DOJ was also allowed to use that report, submitted in one test case, as general causation evidence in other test cases.
None of the petitioners in the test cases could cross-examine Zimmerman, because he was no longer a witness. This was only possible because the federal rules of evidence do not apply in NVICP proceedings.
Yates’ case, Hazlehurst v. HHS, was the second test case in the OAP. His treating neurologist, Dr. Jean-Ronel Corbier testified Yates’ autism was likely caused by a genetic predisposition combined with an environmental insult in the form of vaccinations administered when Yates was ill. (Yates was a patient of Zimmerman in 2002.)
Corbier’s theory of causation in Yates was similar to the theory developed by Zimmerman in the Poling paper and shared with DOJ attorneys.
Yet, despite knowing Zimmerman had concluded that in a subset of children like Yates, vaccines can cause autism, the DOJ “intentionally and fraudulently” misrepresented Zimmerman’s expert testimony in its closing statements in Yates’ case, Hazlehurst alleges.
DOJ attorneys selectively quoted Zimmerman’s expert report from the Cedillo case, telling the court that Zimmerman found there was “no sound evidence to support a causative relationship with exposure to both or either MMR and/or mercury,” when Zimmerman had explicitly told the DOJ that his opinion was the opposite, according to the affidavit.
Fraud #2: the Hannah Poling case
Three weeks after closing arguments in Yates’ case, the DOJ quietly conceded Hannah Poling’s case, which was on the verge of becoming the fourth test case.
Hannah regressed into autism over several months after being vaccinated against nine diseases at one doctor’s visit.
In 2003, Poling’s father, Jon, a physician and trained neurologist, and mother, Terry, an attorney and nurse, filed an autism petition against HHS under the NVICP for their daughter’s injuries.
Jon Poling was a co-author of the 2006 paper with Zimmerman that analyzed an unnamed child, later revealed as Hannah Poling, who had a mitochondrial disorder — a condition with which Yates was later diagnosed.
In 2007, just three weeks after the lead DOJ attorney misrepresented Zimmerman’s opinion during the hearing in Hazlehurst, the same DOJ attorney submitted a report to the special masters conceding that in the case of Poling v. HHS, Hannah’s “regressive encephalopathy with features of autism spectrum disorder” (i.e., regressive autism) was caused by a vaccine injury, based upon a preponderance of the evidence standard.
This was the same neurological diagnosis Zimmerman had made for Yates in 2002.
According to court documents, if HHS had not conceded Poling, Poling v. HHS would have been designated as a test case. However, because the DOJ conceded the case, it was taken out of the omnibus and the DOJ had the case records sealed —- although they were later leaked to the press and published in the Huffington Post in 2008.
In March 2008, Hannah’s parents moved to make the proceedings transparent and available to the public, but the DOJ opposed the motion and the NVICP deferred a ruling on the motion for 60 days.
During those 60 days, the DOJ filed amendments to its report conceding the Poling case. It retroactively changed the basis for compensation to say that Hannah had a “table injury.”
This meant that instead of conceding that the petitioners had proven with a preponderance of evidence that the vaccines caused her autism, they said she had a presumptive injury on the vaccine table, in which causation is presumed.
By conceding the Poling case, opposing the parents’ motion for complete transparency and changing the basis for compensation, the DOJ was able to conceal fraud and critical material evidence of how vaccines cause autism, according to Hazlehurst.
Fraud #3: appellate courts and the U.S. Supreme Court
On Feb. 12, 2009, the special masters denied compensation in the first three cases. They found the petitioners failed to establish causation between MMR or TCV vaccines and autism.
In Hazlehurst’s case, the NVICP explicitly relied on the portion of Zimmerman’s expert report that DOJ attorneys misrepresented.
The Hazlehursts appealed to the Court of Federal Claims and the Court of Appeals for the Federal Circuit, both of which upheld the special master’s decision — by relying on Zimmerman’s misrepresented opinion and knowingly fraudulent statements made by a DOJ attorney, according to Hazlehurst.
Those prior decisions directly influenced the U.S. Supreme Court’s decision in the Bruesewitz v. Wyeth.
In that case, Wyeth, now Pfizer, argued that a decision favoring the Bruesewitz family — who was attempting to sue the company for their daughter’s vaccine injury — would lead to a “flood of frivolous lawsuits,” including by the families from the omnibus.
Amicus briefs from the American Academy of Pediatrics, GlaxoSmithKline, Merck and Sanofi Pasteur on behalf of Wyeth relied on Hazlehurst v. HHS and other OAP decisions that were based on the misrepresentation of Zimmerman’s testimony that there was “no scientific basis” that vaccines cause autism.
The Supreme Court ruled that the National Childhood Vaccine Injury Act, and the NVICP it created, preempt all design-defect claims against vaccine manufacturers by individuals seeking compensation for injury or death.
In oral arguments and in their written opinions, the justices explicitly cited the portions of the amicus briefs citing Hazlehurst v. HHS and other OAP rulings that relied on the DOJ misrepresentations in their rulings.
Since that ruling, the special masters have continued to rely on the DOJ’s fraudulent claims to deny compensation to families filing complaints in the NVICP.
Robert F. Kennedy Jr., CHD chairman on leave, and Hazlehurst in September 2018 filed a complaint with the DOJ Office of Inspector General outlining what they then knew about the DOJ’s fraud during the OAP.
The DOJ Office of Professional Misconduct investigated and responded in a June 2019 letter that it found no wrongdoing.
In that letter, however, the Office of Professional Responsibility conceded the DOJ had in fact kept Zimmerman’s testimony while dismissing him as a witness in order to avoid creating the appearance that he had changed his opinion and to prevent the petitioners from cross-examining him, according to Hazlehurst.
The ‘fraud on the court’ doctrine
It has taken 17 years, Hazlehurst said, since the DOJ’s first alleged act of fraud upon the court, for him to gather all of the admissible evidence necessary to “connect the dots and reveal the DOJ’s web of deceit” to make this claim under the “fraud on the court” doctrine.
Under this doctrine, codified as Rule 60(d)(3) in the rules of the Court of Federal Claims, there is no time limit for the court to overturn a judgment made on the basis of fraud on the court.
The petitioner must demonstrate that there was fraud, intent to defraud and that the fraud affected more than one instance of litigation — putting the integrity of the judicial process at stake.
Hazlehurst alleges DOJ attorneys committed fraud by knowingly making false statements and offering evidence they knew to be false and that they did not take remedial action to disclose information they knew to be false and misleading to the court.
The special masters themselves have an obligation to consider all relevant evidence, but didn’t, in this case, Hazlehurst said. Instead, they ignored the contradictions in Zimmerman’s opinions and ignored the Poling evidence.
This is particularly problematic for NVICP cases, where petitioners can’t conduct meaningful discovery or cross-examination and the special masters’ oversight is the only meaningful safeguard to prevent the DOJ’s abuse of power, according to Hazlehurst.
“There is nothing fair about a government proceeding where the government controls the admissibility of evidence,” he said.
Hazlehurst said that by forcing people injured by vaccines into an administrative program, petitioners are deprived of the basic constitutional rights to due process and equal protection under the law. “It should be declared unconstitutional,” he said.
Brenda Baletti Ph.D. is a reporter for The Defender. She wrote and taught about capitalism and politics for 10 years in the writing program at Duke University. She holds a Ph.D. in human geography from the University of North Carolina at Chapel Hill and a master’s from the University of Texas at Austin.
This article was originally published by The Defender — Children’s Health Defense’s News & Views Website under Creative Commons license CC BY-NC-ND 4.0. Please consider subscribing to The Defender or donating to Children’s Health Defense.
Big Pharma designed WHO’s Global Health Policy from 2000-2009
Corruption and deception, not science, is the foundation of WHO health policy
By Judy Wilyman PhD | Vaccination Decisions | April 1, 2024
“The past was erased, the erasure was forgotten, the lie became truth.” – George Orwell, 1984
The history of the GAVI alliance, a board that influences the direction and design of WHO’s global health policies, illustrates how these policies have been directly influenced by industry partners from 2000-2009, and not by an objective board selected by the WHO.
This direct influence was hidden from the public in 2009 when the alliance became known as the Gavi board. At this time its composition and function changed to hide the role that industry had played from 2000-2009 in changing the direction of global health policies to a new focus on vaccine production and global implementation.
History of the Gavi Board:
In 1998 the Global Alliance for Vaccines and Immunisation (GAVI) was established by the Head of the World Bank after a meeting with pharmaceutical companies and other agencies. The GAVI alliance was established on the advice of industry because the pharmaceutical companies were claiming that there was no incentive for them to provide vaccines to the developing countries.
This meeting led to the Bill and Melinda Gates Foundation providing the seed funding of $750 million in 1999 and governments then matched this figure to establish an alliance of private-public partnerships in 2000, to fund the vaccination programmes for all countries.
In 2000 the alliance was launched at the World Economic Forum (WEF), not the World Health Organisation (WHO), and it established a working party to work with the WHO to design the International Health Regulations (IHR), yet it was a body established outside of the WHO’s charter.
At this time all stakeholders in the Global Alliance for Vaccines and Immunisation (GAVI) were able to directly influence the design of the WHO’s Global Health Policies through this working party (2000-2009), including the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA). They could attend meetings and present information for policy development.
Other stakeholders in the GAVI at this time included the BMGF, the Rockefeller Foundation, the World Bank and the International Monetary Fund (IMF). The influence of these stakeholders led to a new focus on vaccine production and implementation in the WHO’s global health policies.
These global policies were presented to countries in the International Health Regulations (IHR) that came into force in June 2007.
This direct influence of all stakeholders changed in 2009 when the GAVI alliance became known as the GAVI board. Its composition was changed to include only four permanent board members – UNICEF, BMGF, the World Bank and WHO – and other partners would be on a part-time basis.
This change to only four permanent board members, one of which was now the WHO, hides the fact that from 2000-2009 all stakeholders were able to directly influence the design of WHO’s global health policies.
The first recorded meeting of the Gavi board on its website is in 2009. It describes the role of the Gavi board as ‘being responsible for strategic direction and policy-making, oversees the operations of the Vaccine Alliance and monitors programme implementation’ .
This alliance of partners, many of whom profit from vaccines, make donations to the Gavi board and still influence global health policies in a more indirect fashion.
The WHO’s IHR are currently being amended with strong influence from this corporate alliance. If the amendments are approved, the draconian directives implemented during the COVID ‘pandemic’ years, will become binding on every WHO member country, whenever the director of the WHO declares another pandemic. This is removing fundamental human rights and objective scientific evidence from global health policies.
It is time for Australians to make our voices heard to ensure that Australia exits the WHO and joins the World Council for Health to protect both human health and fundamental human rights in all public health policies.
[The information above can be referenced from Ch 3 of my PhD 2015]
Important Information:
- Here is the witness statement from ex-Qantas pilot, Captain Graham Hood, describing that lack of evidence for safety and efficacy that was used by the Australian Therapeutic Goods Administration (TGA) and the Australian Prime Minister, Scott Morrison, to mandate this mRNA genetically-engineered injection (called a ‘vaccine’) in the Australian population – Ex-Qantas Pilot, Graham Hood, provides a witness statement in the Australian parliament.
- Australian Medical Professional Society (AMPS) presents ‘Too Many Dead’ in Australia, but the Australian government will not investigate and the media does not report these facts.
- Study finds the Majority of Patients with Long COVID were Vaccinated
- Epidemic of Fraud
Do the Pertussis Vaccines Used in the US Stop Infection and Transmission of the Pertussis Bacterium?
Your bite-size dose of immunity against vaccine misinformation. Spread the truth.
Injecting Freedom by Aaron Siri | March 22, 2024
Do the pertussis (whooping cough) vaccines used in the United States stop infection and transmission of the pertussis bacterium?
“Yes” or “No”?
When picking an answer, keep in mind that the pertussis vaccine is part of a combination vaccine (DTap or Tdap) mandated to attend grades K-12 in every U.S. state – it is the “P” in DTaP and the “p” in Tdap – and the justification for this rights-crushing mandate is the belief that the vaccine prevents transmission of pertussis in the school setting.
The answer is “No”! In 1999, the CDC recommended “exclusive use of acellular pertussis vaccines for all doses of the pertussis vaccine series” and that vaccine does not prevent transmission. This is explained in an FDA study titled “Acellular pertussis vaccines protect against disease but fail to prevent infection and transmission in a nonhuman primate model” and confirmed in a consensus paper explaining that:
“aPVs [pertussis vaccines] … cannot avoid infection and transmission. … aPV pertussis vaccines do not prevent colonization. Consequently, they do not reduce the circulation of B. pertussis and do not exert any herd immunity effect.”
The CDC and FDA, in formal responses to the Informed Consent Action Network (ICAN), confirm the foregoing, as does this paper explaining:
“That vaccination does not prevent B. pertussis infection in humans, nor the circulation of the organism in human populations in any important manner, comes from the observation that the inter-epidemic intervals have not changed in a major way since the implementation of mass vaccination.”
Incredibly, the immunity provided by pertussis vaccines, while potentially reducing symptoms of the disease, actually renders those receiving these products susceptible to repeated infection with pertussis; meaning, it increases the potential to spread this bacterium because it renders those vaccinated repeat-asymptomatic-carriers. (See this study, “Lack of mucosal immune responses after aPV administration favor infection, persistent colonization, and transmission of the pathogen”, and this study, “Because of linked-epitope suppression, all children who were primed by DTaP vaccines will be more susceptible to pertussis throughout their lifetimes, and there is no easy way to decrease this increased lifetime susceptibility.”)
In any event, immunity from the pertussis vaccine wanes rapidly, even after six doses in childhood! As the CDC explains, a study of pertussis vaccine immunity found that four years after five doses of DTaP and one of Tdap “vaccine effectiveness was 8.9%.” Nonetheless, the CDC makes additional doses of the pertussis vaccine optional in adulthood.
Screenshots of the relevant portions of the websites linked above can be viewed here (in case they change).
Is Russia sending the WHO a shot across the bow?
“Thinning of humanity,” lack of accountability and WHO coverups are all mentioned in a few sentences. What might this portend?
BY MERYL NASS | MARCH 28, 2024
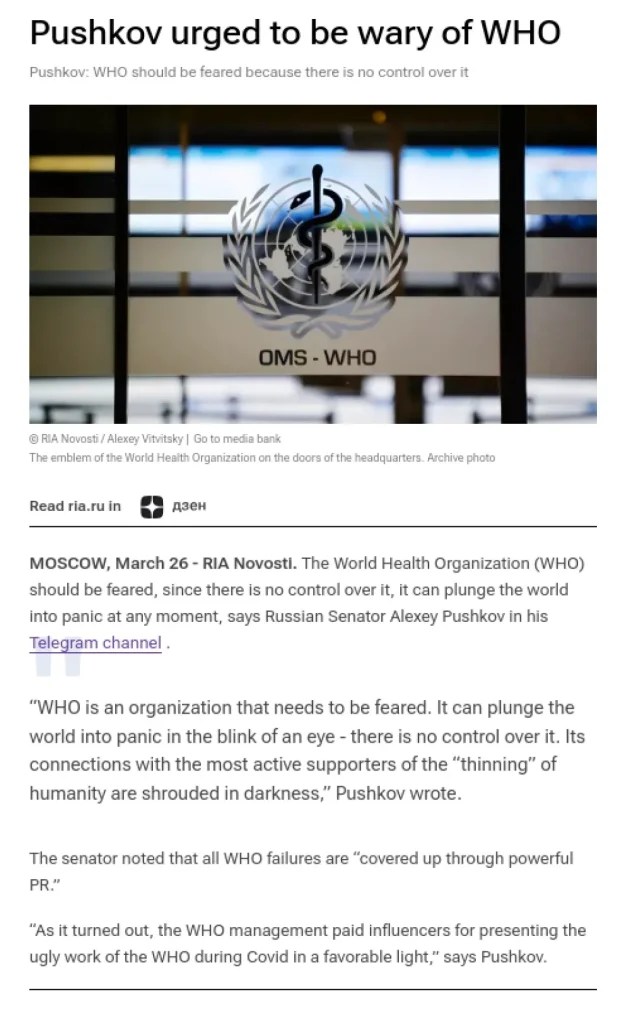
 Russian Senator Aleksey Pushkov took to Telegram to complain about the WHO, and then RIA Novosti reported on it.
Russian Senator Aleksey Pushkov took to Telegram to complain about the WHO, and then RIA Novosti reported on it.
What is RIA Novosti ? “RIA Novosti is the most cited Russian news agency in the mass media and across social media.”
Who is Pushkov? Below is his bio; but important to note that he is also claimed to be a good friend of Vladimir Putin.
Aleksey Konstantinovich Pushkov, born 10 August 1954, is a Russian politician who has been Senator from Perm Krai since 29 September 2016. He is also a former Deputy of the State Duma and former head of the Foreign Affairs Committee in the State Duma, the lower house of the Russian Parliament. As a member of the United Russia political party in the federation council, he is the chairman of the Commission on Information Policy.
What did Pushkov say, and how was it reported? Here is what RIA reported in English:
FDA is trying to legalize its illegal approvals of COVID vaccines without human trials
Now one animal trial will do the trick to “prove” efficacy?

BY MERYL NASS | MARCH 26, 2024
You can’t prove efficacy with an animal trial. You can’t prove safety with an animal trial. The anthrax vaccine studies PROVED that even when a vaccine seemed to work in animals, it was unclear what the right dose was and equally unclear how antibody levels related to protection, because they didn’t. Maybe because no one knew which antibody to measure?
Surrogate markers for approval of vaccines (or anything else) became a total joke when FDA began ignoring the requirement that “correlates of protection” were a essential before you could use antibodies, T cells, animals or anything besides humans to assess a vaccine. This means that you have to prove the surrogate marker or animal model actually duplicates the human response, since usually they don’t.
FDA officials ditched this absolutely necessary standard (imho illegally) and now want to a) cover their tracks, and b) foist more untested and possibly deadly injections on us going forward.
Who needs them? They are simply a Potemkin Village of a regulatory agency at this point. Thankfully ICAN is watching.
With public faith in FDA continuing to decline, the agency published draft guidance stating that a single animal study could be sufficient to demonstrate a vaccine’s effectiveness. ICAN’s attorneys submitted a comment in opposition to this draft guidance which comes shortly after FDA approved a COVID-19 booster based on trials performed solely in mice.
Back in 2007, an FDA advisory committee met to re-review the safety, effectiveness, and dosing of phenylephrine (Sudafed PE), a drug which has been on the market for nearly 50 years. At the 2007 meeting, the committee considered its efficacy data “borderline” but nonetheless voted 11-1 in favor of its efficacy, although it did vote in favor of additional studies being done.
On September 12, 2023, the same FDA committee, possibly attempting to bolster its image as a tough regulatory watchdog, voted unanimously to label phenylephrine “ineffective” as a nasal decongestant. The committee noted “significant methodological and statistical issues with the design and conduct of the original studies submitted to and evaluated by the Panel… All but one evaluated extremely small sample sizes, none adequately controlled for bias … and none performed appropriate sample size calculations,” and observed, “After a thorough review of all the available evidence, it is also possible that there may have been bias and/or data integrity issues at [at] least one study center … where five of the seven positive oral PE studies were conducted.”
If FDA follows the panel’s advice, the drug will be pulled from the market, despite the long-standing indications of its inefficacy, most of which were known in 2007. It’s too bad the committee didn’t perform this “thorough review” at any point prior to the last 50 years, before Americans wasted billions of dollars on an ineffective treatment (nearly $2 billion in 2022 alone) that apparently only exposed them to potential harm without benefit.
Ironically, on September 11, 2023, just one day before the committee met to clamp down on this “ineffective drug” with shoddy clinical trials, FDA issued draft guidance stating that, under certain circumstances, a manufacturer can show evidence of effectiveness for a biological product with a single clinical investigation conducted in animals, giving the example of “[w]hen the product is a preventive vaccine, and there is a well-established model of infection for a relevant infectious disease, and use of the vaccine in the animal model demonstrates prevention of disease.” Notably, this occurred shortly after FDA authorized the newest Pfizer COVID-19 vaccines, which are as Dr. Paul Offit notes, “new product[s],” even though Pfizer tested this brand-new shot only in mice — not humans.
Thus, on December 18, 2023, ICAN, through its attorneys, filed a formal comment in opposition to the draft guidance. In it, ICAN points out that, despite urging by ICAN in its November 2020 petition, FDA refused to amend the Phase III trials of the COVID-19 vaccines to ensure they met the required standard of “substantial evidence” of effectiveness. As a result, we now have generations of new vaccines being approved based on those original ineffective products.
Now, FDA is doubling down on its malfeasance by authorizing COVID-19 vaccines without human trials while simultaneously creating guidance to excuse its lack of oversight. As ICAN’s comment notes:
[I]t is apparent that FDA is tailoring guidance based on the vaccine manufacturers’ clinical trials instead of requiring that these trials comply with what any reasonable licensing agency should and would require. FDA has seemingly forgotten that its function is to regulate the pharmaceutical industry, not rubber stamp it. The Draft Guidance does not provide oversight, and even worse lends illegitimacy to the FDA when there is clearly insufficient evidence to support authorization or licensure. This is especially troubling for products that will be injected into healthy humans including babies, children, and pregnant women.
Rest assured that ICAN’s legal team will continue to serve as a watchdog for FDA and ensure it is held accountable for improperly wielding its regulatory authority and fulfilling its responsibilities.
To support future legal action like this, click here to donate!
You can read more about ICAN’s work serving as watchdog for our federal health agencies at the links below: