Pfizer Vaccine Clinical Trials Poorly Designed From the Start, Analysis Shows
By Madhava Setty, M.D. | The Defender | December 22, 2021
The disagreements around whether COVID-19 vaccinations are “safe and effective” stem not only from the subjective nature of such descriptors but also from the lack of consensus around the data used to substantiate or refute such a claim.
The Centers for Disease Control and Prevention’s (CDC) Morbidity and Mortality Weekly Reports (MMWR) often draw from limited observations, and their assessment of vaccine effectiveness is based on relatively small subsets of our population.
Their conclusions are sometimes reflective of outdated data and conflict with their own recommendations.
For example, here the CDC’s latest estimation of vaccine effectiveness is upwards of 80% in preventing COVID-19 (i.e. the risk of becoming infected is 5 times less if vaccinated). Why then the recommendation for boosters?
Note that this latest metric is based on the CDC’s most recent data from more than one month ago and represents data drawn only from 27 jurisdictions.
Eric Topol, a professor of molecular medicine at Scripps Research, is a former advisory board member of the Covid Tracking Project, a team that worked to collect and synthesize local COVID-19 during the peak of the pandemic.
Politico recently quoted Topol:
“I think we’ve done a horrible job from day one in data tracking for the pandemic. We’re not tracking all the things that we need to to get a handle on what’s going on. It is embarrassing.”
Meanwhile, vaccine injuries continue to accumulate in the Vaccine Adverse Event Reporting System (VAERS) and go unacknowledged, making any constructive discussion around risk impossible.
In order to find common ground, it is perhaps most appropriate to focus on published data from the vaccine trials themselves.
Was Emergency Use Authorization justified? Was the current confusion around vaccine effectiveness and safety predictable from the beginning?
A presentation by the Canadian COVID Care Alliance
Canadian COVID Care Alliance (CCCA) is a group of “Independent Canadian doctors, scientists and healthcare professionals committed to providing top quality and balanced evidence-based information to the Canadian public about COVID-19 so that hospitalizations can be reduced, lives saved and our country restored as safely as possible.”
CCCA assembled a presentation that comprehensively demonstrates how Pfizer’s purported randomized placebo-controlled, double-blinded study veered away from methodologies that would have answered the safety and efficacy questions definitively.
In this concise slide deck with an explanatory video, CCCA powerfully summarized why Pfizer’s trial was not designed to adequately demonstrate its product’s safety and efficacy.
Here are a few key points from the CCCA presentation:
- Initial data demonstrated a high relative risk reduction of infection yet this amounted to an absolute risk reduction of only 0.84%. It is the absolute risk reduction that determines the risk-benefit ratio required to make informed decisions around inoculation.
- Early unblinding: Several months before publishing six-month observational results Pfizer opted to offer its product to those participants who received the placebo. By eliminating nearly all participants in the placebo wing Pfizer effectively closed the curtain on its experiment because long-term comparisons can no longer be made.
- All-cause mortality and morbidity, the only sensible outcomes to use in determining efficacy and risk, were not considered. Indeed, all-cause mortality was higher in the vaccinated group after six months.
- Severe adverse events outnumbered cases of severe COVID prevented after six months of observation.
- Trial participants were not reflective of the most vulnerable members of our population — more than 50% of people dying from COVID are 75 years of age or older. This age group made up only 4.4% of trial participants. Also, 95% of those who have died from COVID had one or more comorbidities. Nearly 80% of trial participants had none.
- Not every trial participant was tested for COVID. Asymptomatic or paucisymptomatic (presenting few symptoms) cases were missed.
Questions regarding unblinding and data integrity
The CCCA presentation also resurrects a puzzling observation mentioned in a briefing document Pfizer submitted only to the FDA’s Vaccine and Related Biologic Products Advisory Committee (VRBPAC) of the FDA, but nowhere else — including the widely cited summary of the trial reported in New England Journal of Medicine.
According to the document, 3,410 participants were suspected from their clinical presentation of having COVID but they were excluded from efficacy calculations because a diagnosis could not be confirmed through PCR testing.
The CCCA presentation presumes this large group of participants was never tested. The wording in the VRBPAC briefing document is indeed vague, stating the participants were “not PCR-confirmed” in one sentence and “unconfirmed” in another.
Assuming Pfizer’s investigators followed their study protocol, these participants were in fact tested. Yet that forces us to accept that more than 3,400 participants who had symptoms of COVID were suffering from other illnesses, not COVID.
In other words, there were 3,580 participants who clinically presented with COVID (3,410 suspected and 170 confirmed). Of these, more than 95% tested negative. This is difficult to accept in a group where clinical suspicion is high.
However, with no further testing by the investigators, we are left to accept these numbers as reported.
Peter Doshi, Ph.D., senior editor at The BMJ, explained the implications of this result in detail, in an opinion piece published nearly one year ago.
In his widely discussed commentary, Doshi noted another baffling finding in the Pfizer data. Within 7 days of administration of the second of two doses, 371 (310 in the vaccinated group and 61 in the placebo group) trial participants were withdrawn from the study due to “important protocol deviations.”
Of course, protocol deviations occur, but why were five times more vaccine recipients excluded than placebo recipients at that point in the study?
Although there were nearly 40,000 participants in the evaluable population, only 170 contributed to the efficacy calculation with regard to protection from infection, and only 10 with regard to protection from severe infection.
In other words, just a handful of incorrectly diagnosed and categorized participants could easily result in a substantially different estimation of the vaccine’s efficacy and safety.
Statistician and educator Mathew Crawford pointed out the likelihood of such a disparity between groups is exceedingly unlikely. However, because the investigators should have been blinded, we must accept this as an extraordinary coincidence.
Incredibly, the very same disparity occurred in the pediatric trials (ages 5 to 11). Table 12 from the corresponding summary to the FDA’s VRBPAC indicated 3.1% of children were removed from the trial if they received the vaccine compared to 0.5% if they received the placebo.
Once again, there is a factor of 5 difference at exactly the same point in the trial (within 7 days of dose 2). It is true that the vaccine formulation requires steps prior to administration that are not required with the placebo, but why didn’t these protocol deviations happen with the first dose as well? What happened to these vaccine recipients soon after they received their second dose?
These remarkable “coincidences” can be best explained if we are willing to consider the possibility that the investigators were not, in fact, blinded. This is precisely the most damning allegation by whistleblower Brook Jackson, formerly a regional director at Ventavia, one of several clinical research organizations conducting Pfizer’s vaccine trials in 2020.
In addition to the unblinding of investigators, Jackson also accused Ventavia of falsifying data.
These are weighty accusations. Why should we believe her? Because her story is corroborated by the trial data themselves.
She was fired within a day of reporting her concerns directly to the FDA. Her story was covered in The BMJ Nov. 2. Mainstream media has yet to report on it.
Conclusion
Though public health authorities continue to proclaim these products are safe and effective, every week brings more and more evidence to the contrary.
A thorough analysis of data from Pfizer’s vaccine trials reveals more questions than answers. Claims the vaccine maker’s product is performing “as expected” may not be so far from the truth.
© 2021 Children’s Health Defense, Inc. This work is reproduced and distributed with the permission of Children’s Health Defense, Inc. Want to learn more from Children’s Health Defense? Sign up for free news and updates from Robert F. Kennedy, Jr. and the Children’s Health Defense. Your donation will help to support us in our efforts.
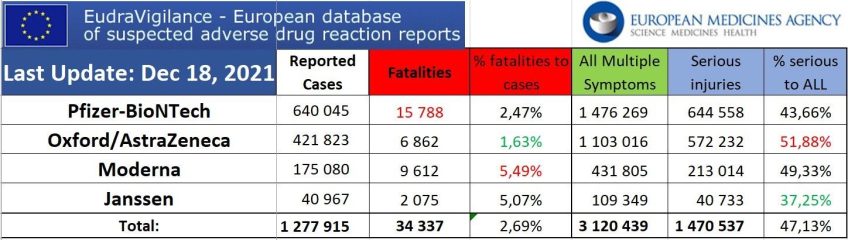
34,337 Deaths 3,120,439 Injuries Following COVID Shots in European Database
UK Public Data Show 35 Deaths 213 Hospitalizations Among Booster Triple Vaccinated

By Brian Shilhavy | Health Impact News | December 22, 2021
The European (EEA and non-EEA countries) database of suspected drug reaction reports is EudraVigilance, verified by European Medicines Agency (EMA), and they are now reporting 32,649 fatalities, and 3,003,296 injuries following injections of four experimental COVID-19 shots:
- COVID-19 MRNA VACCINE MODERNA (CX-024414)
- COVID-19 MRNA VACCINE PFIZER-BIONTECH
- COVID-19 VACCINE ASTRAZENECA (CHADOX1 NCOV-19)
- COVID-19 VACCINE JANSSEN (AD26.COV2.S)
From the total of injuries recorded, almost half of them (1,470,537) are serious injuries.
“Seriousness provides information on the suspected undesirable effect; it can be classified as ‘serious’ if it corresponds to a medical occurrence that results in death, is life-threatening, requires inpatient hospitalisation, results in another medically important condition, or prolongation of existing hospitalisation, results in persistent or significant disability or incapacity, or is a congenital anomaly/birth defect.”
A Health Impact News subscriber in Europe ran the reports for each of the four COVID-19 shots we are including here. It is a lot of work to tabulate each reaction with injuries and fatalities, since there is no place on the EudraVigilance system we have found that tabulates all the results.
Since we have started publishing this, others from Europe have also calculated the numbers and confirmed the totals.*
Here is the summary data through December 18, 2021.
Total reactions for the mRNA vaccine Tozinameran (code BNT162b2,Comirnaty) from BioNTech/ Pfizer: 15,788 deaths and 1,476,269 injuries to 18/12/2021
- 40,271 Blood and lymphatic system disorders incl. 226 deaths
- 47,256 Cardiac disorders incl. 2,310 deaths
- 433 Congenital, familial and genetic disorders incl. 41 deaths
- 19,912 Ear and labyrinth disorders incl. 11 deaths
- 1,504 Endocrine disorders incl. 5 deaths
- 22,804 Eye disorders incl. 35 deaths
- 120,263 Gastrointestinal disorders incl. 637 deaths
- 370,545 General disorders and administration site conditions incl. 4,452 deaths
- 1,691 Hepatobiliary disorders incl. 82 deaths
- 16,314 Immune system disorders incl. 84 deaths
- 61,494 Infections and infestations incl. 1,649 deaths
- 25,540 Injury, poisoning and procedural complications incl. 279 deaths
- 36,772 Investigations incl. 476 deaths
- 10,065 Metabolism and nutrition disorders incl. 264 deaths
- 179,558 Musculoskeletal and connective tissue disorders incl. 196 deaths
- 1,362 Neoplasms benign, malignant and unspecified (incl cysts and polyps) incl. 128 deaths
- 246,596 Nervous system disorders incl. 1,694 deaths
- 2,127 Pregnancy, puerperium and perinatal conditions incl. 64 deaths
- 223 Product issues incl. 3 deaths
- 26,890 Psychiatric disorders incl. 191 deaths
- 5,314 Renal and urinary disorders incl. 249 deaths
- 55,551 Reproductive system and breast disorders incl. 5 deaths
- 63,512 Respiratory, thoracic and mediastinal disorders incl. 1,742 deaths
- 68,837 Skin and subcutaneous tissue disorders incl. 134 deaths
- 3,257 Social circumstances incl. 21 deaths
- 10,192 Surgical and medical procedures incl. 122 deaths
- 37,986 Vascular disorders incl. 688 deaths
Total reactions for the mRNA vaccine mRNA-1273(CX-024414) from Moderna: 9,612 deaths and 431,805 injuries to 18/12/2021
- 9,176 Blood and lymphatic system disorders incl. 106 deaths
- 14,538 Cardiac disorders incl. 1,000 deaths
- 174 Congenital, familial and genetic disorders incl. 8 deaths
- 5,244 Ear and labyrinth disorders incl. 4 deaths
- 409 Endocrine disorders incl. 4 deaths
- 6,337 Eye disorders incl. 33 deaths
- 35,162 Gastrointestinal disorders incl. 359 deaths
- 114,485 General disorders and administration site conditions incl. 3,239 deaths
- 693 Hepatobiliary disorders incl. 47 deaths
- 4,314 Immune system disorders incl. 17 deaths
- 16,686 Infections and infestations incl. 907 deaths
- 8,851 Injury, poisoning and procedural complications incl. 180 deaths
- 8,917 Investigations incl. 263 deaths
- 4,138 Metabolism and nutrition disorders incl. 231 deaths
- 51,645 Musculoskeletal and connective tissue disorders incl. 193 deaths
- 595 Neoplasms benign, malignant and unspecified (incl cysts and polyps) incl. 76 deaths
- 72,360 Nervous system disorders incl. 919 deaths
- 785 Pregnancy, puerperium and perinatal conditions incl. 6 deaths
- 91 Product issues incl. 4 deaths
- 7,887 Psychiatric disorders incl. 158 deaths
- 2,553 Renal and urinary disorders incl. 183 deaths
- 9,972 Reproductive system and breast disorders incl. 8 deaths
- 19,269 Respiratory, thoracic and mediastinal disorders incl. 1,032 deaths
- 23,101 Skin and subcutaneous tissue disorders incl. 84 deaths
- 1,956 Social circumstances incl. 40 deaths
- 1,946 Surgical and medical procedures incl. 150 deaths
- 10,521 Vascular disorders incl. 361 deaths
Total reactions for the vaccine AZD1222/VAXZEVRIA (CHADOX1 NCOV-19) from Oxford/ AstraZeneca: 6,862 deaths and 1,103,016 injuries to 18/12/2021
- 13,469 Blood and lymphatic system disorders incl. 255 deaths
- 19,919 Cardiac disorders incl. 753 deaths
- 208 Congenital familial and genetic disorders incl. 7 deaths
- 13,018 Ear and labyrinth disorders incl. 5 deaths
- 642 Endocrine disorders incl. 4 deaths
- 19,414 Eye disorders incl. 30 deaths
- 104,504 Gastrointestinal disorders incl. 370 deaths
- 289,568 General disorders and administration site conditions incl. 1,614 deaths
- 985 Hepatobiliary disorders incl. 63 deaths
- 5,105 Immune system disorders incl. 31 deaths
- 34,377 Infections and infestations incl. 471 deaths
- 12,816 Injury poisoning and procedural complications incl. 184 deaths
- 24,316 Investigations incl. 178 deaths
- 12,629 Metabolism and nutrition disorders incl. 101 deaths
- 163,096 Musculoskeletal and connective tissue disorders incl. 125 deaths
- 684 Neoplasms benign malignant and unspecified (incl cysts and polyps) incl. 29 deaths
- 226,199 Nervous system disorders incl. 1,047 deaths
- 558 Pregnancy puerperium and perinatal conditions incl. 17 deaths
- 193 Product issues incl. 1 death
- 20,437 Psychiatric disorders incl. 62 deaths
- 4,164 Renal and urinary disorders incl. 66 deaths
- 15,992 Reproductive system and breast disorders incl. 2 deaths
- 39,444 Respiratory thoracic and mediastinal disorders incl. 880 deaths
- 50,458 Skin and subcutaneous tissue disorders incl. 54 deaths
- 1,563 Social circumstances incl. 6 deaths
- 1,611 Surgical and medical procedures incl. 29 deaths
- 27,647 Vascular disorders incl. 478 deaths
Total reactions for the COVID-19 vaccine JANSSEN (AD26.COV2.S) from Johnson & Johnson: 2,075 deaths and 109,349 injuries to 18/12/2021
- 1,068 Blood and lymphatic system disorders incl. 44 deaths
- 2,052 Cardiac disorders incl. 173 deaths
- 41 Congenital, familial and genetic disorders incl. 1 death
- 1,146 Ear and labyrinth disorders incl. 2 deaths
- 87 Endocrine disorders incl. 1 deaths
- 1,475 Eye disorders incl. 7 deaths
- 8,932 Gastrointestinal disorders incl. 84 deaths
- 28,855 General disorders and administration site conditions incl. 559 deaths
- 138 Hepatobiliary disorders incl. 13 deaths
- 489 Immune system disorders incl. 10 deaths
- 5,092 Infections and infestations incl. 165 deaths
- 1,011 Injury, poisoning and procedural complications incl. 21 deaths
- 5,043 Investigations incl. 115 deaths
- 687 Metabolism and nutrition disorders incl. 51 deaths
- 15,638 Musculoskeletal and connective tissue disorders incl. 47 deaths
- 68 Neoplasms benign, malignant and unspecified (incl cysts and polyps) incl. 6 deaths
- 21,175 Nervous system disorders incl. 224 deaths
- 46 Pregnancy, puerperium and perinatal conditions incl. 1 death
- 29 Product issues
- 1,551 Psychiatric disorders incl. 20 deaths
- 462 Renal and urinary disorders incl. 27 deaths
- 2,485 Reproductive system and breast disorders incl. 6 deaths
- 3,937 Respiratory, thoracic and mediastinal disorders incl. 268 deaths
- 3,370 Skin and subcutaneous tissue disorders incl. 9 deaths
- 358 Social circumstances incl. 4 deaths
- 745 Surgical and medical procedures incl. 61 deaths
- 3,369 Vascular disorders incl. 156 deaths
*These totals are estimates based on reports submitted to EudraVigilance. Totals may be much higher based on percentage of adverse reactions that are reported. Some of these reports may also be reported to the individual country’s adverse reaction databases, such as the U.S. VAERS database and the UK Yellow Card system. The fatalities are grouped by symptoms, and some fatalities may have resulted from multiple symptoms.
Meanwhile, The Exposé is reporting that public health data in the UK shows that the vast majority of deaths and hospitalizations in the UK are among those vaccinated, and now there are 35 deaths and 213 hospitalizations being reported among those who have received booster shots and are triple-vaccinated.
Australia’s new Online Safety Act will fine platforms that don’t remove content when ordered
By Christina Maas | Reclaim The Net | December 22, 2021
Australia has passed a new law that will force Google and other search engines to remove content or risk huge fines.
The Online Safety Act will come into effect on January 23. Sites will have only 24 hours to remove harmful content. The penalty for non-compliance is $110,000 for individuals and $550,000 for companies.
The act also applies for apps on both Android and iOS devices.
The eSafety Commissioner Julie Inman Grant also has the authority to name and shame platforms that do not comply with content takedown requests.
Speaking to The Daily Telegraph newspaper, Inman Grant Said: “There aren’t powers like these anywhere in the world. We will use them judiciously. But we feel emboldened to tackle the worst of the worst content.”
Enforcement will apply to both local and international sites.
The Commissioner said that the focus of the act is content with the potential to cause “serious psychological or physical harm.”
Insulting someone might not meet the threshold, unless it is something that will “do more than hurt a person’s feelings.”
Before eSafety can get involved, someone has to make a complaint to the platform hosting the content.
In 2020, eSafety received about 21,000 complaints, a 90% increase from 2019.
“With these new powers, we will now be able to take real action to disrupt the trade in this distressing material and if online service providers fail to comply with our removal notices, they will face very real and significant consequences,” Inman Grant said.
EU’s top court allows European firms to scrap Iran deals
Press TV – December 22, 2021
The EU’s supreme court has intervened to protect European companies against legal action by Iran for failing to fulfill their contractual obligations.
The European Court of Justice (ECJ) in Luxembourg has ruled that EU companies can end contracts with Iranian firms if upholding the deals would lead to “disproportionate economic loss” as a result of US sanctions.
The ruling was prompted by a lawsuit from the German branch of Iran’s Bank Melli against Deutsche Telekom after the telecommunications provider terminated a contract with the bank in 2018 prior to its expiry.
The decision effectively neutralizes a “blocking statute” that prohibited individuals and companies in the European Union from complying with US sanctions imposed in 2018 by then US president Donald Trump after he decided to withdraw unilaterally from the Iran nuclear deal.
As per their obligations under the nuclear deal, the Europeans issued the statute in order to keep Iran in the agreement, but companies in the bloc quickly severed ties rather than risk running afoul of the US.
Under the blocking statute, European firms must seek a legal exemption for withdrawing from Iran due to US sanctions and those failing to do so could be penalized by their governments. Firms, however, can twist the law if they claim that their withdrawal is a business decision.
The court on Tuesday paid lip service to the EU blocking statute in its ruling, saying “the prohibition imposed by EU law on complying with secondary sanctions laid down by the United States against Iran may be relied on in civil proceedings”.
But the judges also said the rules of the blocking statute “cannot infringe the freedom to conduct a business by leading to disproportionate economic loss”.
The Higher Regional Court in Hamburg will have to decide whether upholding the contract with Bank Melli would expose Deutsche Telekom to such a disproportionate economic loss.
Observers believe the decision is a foregone conclusion, given that Deutsche Telekom makes about half of its turnover with its US business.
The ECJ said the Hamburg judges must take into account that Deutsche Telekom did not apply for an exemption from the EU blocking statute’s rules.
Other European measures taken to maintain open trade channels with Iran have equally proven to be empty shells.
For example, Iranians have got almost nothing from the Swiss Humanitarian Trade Agreement (SHTA) since it was launched in January 2020 with the support and consent of the US.
The Swiss trade channel much publicized by Washington as a purportedly secure way of delivering humanitarian assistance to Iran at a time of sanctions has failed to process even a single deal on Iranian medicine imports.
The channel was meant to find a way around the US sanctions to use Iranian funds deposited abroad to buy food and medicine for the country via the Swiss bank BCP.
However, companies seeking to participate in the scheme have found it very difficult to comply with the criteria set by the US government to avoid violating the general rules governing the sanctions, said the report.
Fabian Maienfisch, a spokesman for Switzerland’s State Secretariat for Economic Affairs (SECO), which oversees the channel, has admitted in the past that the initiative had effectively failed to meet its objectives.
Such failures and the ECJ’s ruling prove statements by the Iranian government that the Europeans are disingenuous in dealing with the Islamic Republic.
The ruling comes as Iran and the Europeans continue negotiations in Vienna to find a way to remove the US sanctions.
A possible revival of the agreement would require the European companies to return to Iran and fulfill their obligations, but the EU court decision appears to be intentionally timed to provide them a leeway for further violations.
The US is Gaining a Foothold in Uzbekistan
By Valery Kulikov – New Eastern Outlook – 22.12.2021
To create sustainable groundwork for further expansion into Central Asia, Washington has recently placed particular emphasis on developing relations and cooperation with Uzbekistan.
One of such work areas in this country has been the active opening of “American Corners” in Uzbekistan. It is a US government-supported global network of more than 600 open-access educational centers, already implanted in more than 140 countries, seemingly dedicated to “spreading American culture and American values to every country in the world.” However, created in modern libraries, they are one of the main elements of American soft power. The US Embassy opened an “American Corner” in Qarshi in March 2021; the US Embassy plans to open at least six more such facilities throughout Uzbekistan. It has already allocated over $860,000.
Another area of US expansion in Uzbekistan is USAID’s aspirations to take control of the country’s pharmaceutical industry. To this end, USAID has opened a so-called “Quality Club” in Uzbekistan, which, it says, will promote the development of the pharmaceutical industry and local pharmaceutical manufacturers. According to a US Embassy release, the assistance will consist of discussions on updates, problems, and solutions related to regulating drugs and medical devices in Uzbekistan. US representatives present at the “Quality Club” opening discussed the current state of the Uzbek pharmaceutical industry, the contribution of local medicine producers to the common market, and achievements, obstacles, and development directions in the pharmaceutical industry. The advertising declarations of the US Embassy sounded, as always, noble, unless, of course, one keeps in mind that American charities do not do anything for nothing.
For the sake of objectivity in assessing this event, it should be recalled that there is a rigorous certification in the field of pharmaceutical products. And this, in particular, is clearly illustrated by the pharmaceutical war on vaccines against coronavirus. The United States has done quite a lot to keep the Russian Sputnik V vaccine out of that market. Therefore, it is easy to assume that the result of USAID activities will not be the promotion of Uzbek pharmaceutical products on the American and European markets, but the imposition of imports of American drugs to Uzbekistan and the capture of the Uzbek pharmaceutical market. As for the Uzbek industry, which has shown significant growth in recent years, it is unlikely to survive under pressure from USAID and Western corporations, as multinational corporations do not need competitors.
However, in addition to gaining complete control of Uzbekistan’s pharmaceutical industry, USAID has another goal. And it lies in the expansion through Uzbekistan to the entire EAEU (Eurasian Economic Union ) pharmaceutical industry, given that this Central Asian state has obtained observer status in the Eurasian Union and has already begun to adapt its national standards to EAEU requirements. And, given the importance of the EAEU market, USAID expects to take appropriate positions in the EAEU market through the mediation of Uzbekistan and gain access to the latest pharmaceutical developments in the EAEU.
However, the recently intensified “outreach to Uzbekistan” is being carried out by Washington not only in these directions. For example, recently, in Uzbekistan, there have been active discussions of political and economic partnership between the two countries with the participation of Assistant Secretary of State for South and Central Asian Affairs Donald Lu. The most promising directions of further expansion of the bilateral economic partnership, including mining, chemical, agriculture, textile, and other industries, have been outlined during the meeting held on December 13 in Tashkent. The US side emphasized that in the eleven months of 2021, trade turnover between the two countries increased by 48.5% compared to the same period in 2020. In addition, the number of enterprises with American capital in Uzbekistan has doubled over the past few years. The sides expressed readiness to hold in the first half of 2022 a business forum for representatives of American and Uzbek business communities jointly with the American-Uzbekistan Chamber of Commerce (AUCC).
The US representatives also stressed the importance of strengthening security cooperation by deepening ties between defense, law enforcement, border, and customs agencies. The United States expressed gratitude for the assistance provided by Uzbekistan to humanitarian aid providers at the Termez Cargo Center and welcomed Uzbekistan’s initiative to establish a regional logistics hub in Termez under the auspices of the UN to provide urgent humanitarian aid to the people of Afghanistan.
In the conditions mentioned above, the intensification of military cooperation with Uzbekistan remains on the active agenda of Washington. Uzbekistan remains the most convenient Central Asian country to locate a US Air Force base or counterterrorism center, targeting Afghanistan. Hence, discussions of American and NATO partners with Tashkent continue. Like many post-Soviet republics, Uzbekistan has partnered with NATO for peace since the 1990s, participating in consultations, delegation exchanges, and even joint troop maneuvers on US soil. And yet, for the past 20 years, Uzbek servicemen have not helped the Pentagon in Afghanistan with weapons in their hands like Georgians, Ukrainians and others. On the contrary, closer to the finish line of the infamous US mission in Afghanistan, Tashkent began to successfully establish constructive relations with the Taliban’s “political office” and promote Uzbek-Afghan economic cooperation projects.
Nevertheless, Washington has not given up hope of strengthening the strategic partnership with Uzbekistan in military projects or facilities. The regional choices are too limited. Therefore, representatives of CENTCOM will appear more than once in Tashkent, but the influence of Americans on the situation in the hot region will steadily diminish.
Leaked NHS Report Claims London Hospitals “Likely to Be Overwhelmed” Within Three Weeks. We’ve Heard That Before
By Will Jones | The Daily Sceptic | December 22, 2021
An internal NHS report leaked to the Health Service Journal claims that hospitals and ambulances in London are “likely to become overwhelmed due to rising Covid demand in the next two to three weeks”.
This is despite the fact that, as NHS Providers Chief Chris Hopson points out: “The overall numbers remain relatively low compared to the January 2021 peak – 1,819 Covid patients currently in London versus 7,917 on January 18th 2021.”
Let’s bear in mind also that last winter an internal NHS briefing, again leaked to the Health Service Journal, claimed that under the “best” scenario London would have a shortfall of 1,515 general and acute beds by January 19th. Yet the briefing also stated London has 15,600 general and acute beds, which is almost double what turned out in fact to be the peak Covid winter occupancy of 7,917 beds on January 18th.
Nationally, at the winter peak on January 18th there were 8,696 unoccupied beds, 26,902 beds occupied by Covid patients and 50,204 beds occupied by non-Covid patients.
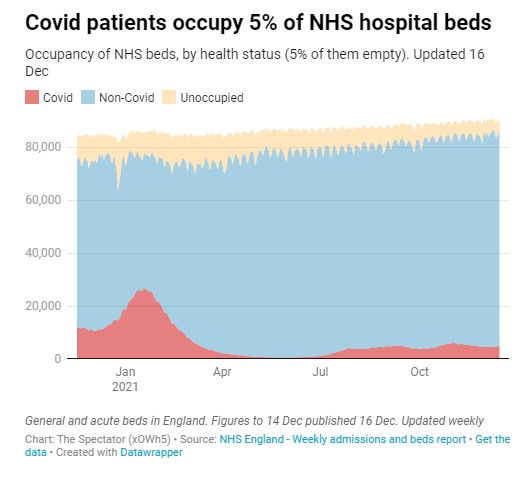
It’s also worth remembering that the NHS has a beds crisis every winter, as this selection of Guardian headlines illustrates. (See also the image at the top of headlines from the 2017-18 winter crisis.)

Yet the NHS coped, just as it did last year when almost no one was vaccinated. If there are systemic capacity problems in the NHS which mean it can’t get through winter without cancelling elective procedures then those need to be addressed. But it should go without saying that this is no excuse to impose costly, illiberal restrictions on society.
Civil liberties should not be so cheap that they may be suspended to make up for the failings of a poorly managed health service with insufficient resources committed to frontline services.
Sen. Johnson Requests Records From Top Medical Journals on Retracted Studies, Including Flawed HCQ Study
The Defender | December 21, 2021
Sen. Ron Johnson (R-Wis.) has written to The Lancet and The New England Journal of Medicine seeking records on two retracted studies from mid-2020. Johnson particularly called out The Lancet study, which suggested hydroxychloroquine could boost the risk of death in COVID patients.
“Although this fraudulent study was ultimately retracted, it is concerning and shameful that, in the midst of a pandemic, The Lancet published such a misleading paper on a potential early treatment for COVID-19,” said Johnson, the ranking member on the Permanent Subcommittee on Investigations, in a letter dated Dec. 14.
Johnson seeks all records of the journals’ communication on the two studies, including communication with the papers’ authors; U.S. government employees; individuals who encouraged the studies’ publication; and the supplier of the two studies’ datasets, Surgisphere, a healthcare analytics company.
Despite The Lancet paper’s retraction, its initial publication halted trials on hydroxychloroquine’s use and sullied its reputation more broadly. The Washington Post and other major media headlined the increased risk of death, and health authorities took action globally within days of the paper’s publication.
The World Health Organization and the UK’s drug regulator halted trials of the drug in COVID settings. France reversed an earlier decision to allow hydroxychloroquine’s use in COVID patients.
Readers of The Lancet quickly noted the study cited implausibly high numbers of COVID cases in 2020, and journalists failed to find any hospitals that had contributed data, despite the study’s claim that more than 96,000 hospital patients participated.
The Lancet retracted the study two weeks after publication.
Sen. Johnson also requested information from The New England Journal of Medicine (NEJM) on another study retracted in June 2020.
Johnson explained in his letter, the NEJM paper reportedly found that “taking certain blood pressure drugs, including angiotensin-converting enzyme (ACE) inhibitors, didn’t appear to increase the risk of death among COVID-19 patients, as some researchers had suggested.”
However, the study’s authors wrote to the NEJM a few weeks after the study was published, acknowledging they could not validate the primary data supporting the study and apologized “to the editors and to readers of the Journal for the difficulties that this has caused.”
Johnson has requested all records by Jan. 4, 2022.
© 2021 Children’s Health Defense, Inc. This work is reproduced and distributed with the permission of Children’s Health Defense, Inc. Want to learn more from Children’s Health Defense? Sign up for free news and updates from Robert F. Kennedy, Jr. and the Children’s Health Defense. Your donation will help to support us in our efforts.