One of my recent posts on the Daily Sceptic was the subject of a ‘fact check‘ by Full Fact, which self-importantly describes itself as “the UK’s independent fact checking organisation” but is in fact funded by Google, Facebook and George Soros, among others, to help them suppress unapproved news and views. Even U.K. broadcasting regulator Ofcom has said it relies on the organisation to tell it what to censor regarding COVID-19, so unfortunately the dog has teeth and can’t just be ignored as one more absurd website with excessive faith in its own infallibility.
The post in question, from September 10th, simply reported on Public Health England’s latest Vaccine Surveillance report, which included infection rates by vaccination status for the previous month so allowed the calculation of an unadjusted estimate of vaccine effectiveness. Full Fact, however, took exception to the idea that vaccine effectiveness can be estimated in this way, because it wasn’t adjusted for confounders. Or used the wrong population data. Or because the article included the (entirely accurate) claim that the PHE report showed higher infection rates in the vaccinated in some age groups. Or because the heading didn’t include ‘caveats’. Or something. In any case, it was ‘incorrect’.
Here follows my correspondence with them, attempting to explain that the factual errors lay entirely in their ‘fact check’, not in my piece.
September 27th 2021
To: The Editor
Incorrect claim that report from Public Health England shows COVID-19 vaccines have “negative effectiveness” in the over-40s: Full Fact correction request
I’m writing to you from Full Fact, the U.K.’s independent fact checking organisation. I have seen an article you published on Friday September 24th so I know you are already aware of a fact check we published earlier last week, but I wanted to send an email to explain why we wrote that fact check.
The article you published on September 10th had the headline “Vaccines Have NEGATIVE Effectiveness in the Over-40s, as Low as MINUS 38%, Shows New PHE Report”
This headline falsely claims that a report from Public Health England (PHE) shows COVID-19 vaccines having “negative effectiveness” in the over-40s. It is not true that the PHE report shows this.
You note in your article that PHE says its data cannot be used on its own as a reliable measurement of vaccine effectiveness. However your headline makes a claim about vaccine effectiveness based on it.
As you will know we have published a fact check on these claims which is available on our website here:
“Vaccines do not raise your risk of catching Covid”
We are asking that you issue a correction on this article in line with the above. We would also ask that you bear this in mind when writing future articles about this data, including the one you published on Friday. We hope our fact check is helpful in this regard.
Please let me know if you’d like to discuss this further.
Many thanks,
Bethan Davies
Policy and Impact Manager
Full Fact
October 5th 2021
Good afternoon,
I just wanted to follow up on an email I sent last week about a fact check we have written on an article you published on 24th September. I will be updating this fact check on our website this week with details of what action we have taken so I wanted to check in with you before I do this. If you are planning to amend this article I’d be very grateful if you could let me know.
Many thanks,
Bethan
October 5th 2021
Dear Bethan
Thank you for your email.
Apologies – I appear to have missed your first email.
As you are aware, I have written in response to your piece ‘fact-checking’ my article of September 10th (here and now also here).
Your piece wrongly implies that people had been confused by PHE’s report as it “seemed to show for the month in question (August 9th to September 5th) that people in their 40s, 50s, 60s and 70s were more likely to test positive for Covid if they had been vaccinated than if they hadn’t”. However, the report doesn’t “seem” to show that, it plainly does show that. Can you explain why your piece attempts to cast doubt on this correct understanding of the data in PHE’s report, and thus misinform the public about the infection risk among vaccinated and unvaccinated people during that month? Will you be amending your piece to ensure it does not confuse or mislead in this way and makes clear that in fact the PHE report does show that vaccinated people in those age groups were more likely to test positive for Covid during that period?
Your piece’s discussion about population estimates is interesting but I hope you will agree that people are entitled to present data and make calculations based on the population data PHE presents in its reports?
You say in your email: “PHE says its data cannot be used on its own as a reliable measurement of vaccine effectiveness.” Those are your words, not theirs. They say: “The vaccination status of cases, inpatients and deaths is not the most appropriate method to assess vaccine effectiveness…” (emphasis added).
However, regardless of what PHE say is the “most appropriate method”, the fact is that vaccine effectiveness is defined as the reduced risk of infection in the vaccinated compared to the unvaccinated (see here). I am clear in the piece that the VE figures given are unadjusted (though they are controlled for age). I explain the limitations of the estimates and address the reason PHE gives for the sample being biased. This is a perfectly valid approach to presenting an estimate of vaccine effectiveness, provided the limitations are clear. It also needs to be kept in mind that studies which do attempt to adjust for various confounders can come with significant problems of their own (see e.g. this and this).
A study in the Lancet published yesterday confirms that vaccine effectiveness has been declining fast against Delta and over time – and that study used data only up to the start of August. This indicates that the VE figures you quote in your ‘fact check’ to counter mine are out of date. The point of estimating unadjusted VE from real-world data is to try to keep up with how vaccines are faring now, not six months ago. We are not trying to denigrate vaccines – that’s why we are sure to make clear their continued effectiveness against serious illness and death. We are only interested in reporting up-to-date factual information about them.
My question for you is why you appear to be attempting to cover over the fact that infection rates in the vaccinated are very high – on PHE data, higher than in the unvaccinated, with the gap increasing week-on-week? Would fact-checking energies not be better spent on those who continue to claim that the vaccines are highly effective against infection, a claim which looks less and less accurate with each passing week?
I would be grateful for confirmation that you have amended your piece to ensure it does not mislead about current infection rates in vaccinated people (according to PHE data) and about the latest vaccine effectiveness estimates.
Kind regards
Will
Will Jones
Associate Editor – Daily Sceptic
October 7th 2021
Dear Will,
Thank you for your response to my email.
We disagree with your point that we have misunderstood the PHE report.
We acknowledge in our fact check that your article mentions PHE’s caveats, but our fact check and the email we sent you initially are related to your headline, which has no caveats in it.
We are happy with information we included on vaccine effectiveness and we have made it clear to readers where this came from.
We very much appreciate you setting out your position. In conclusion however, after consideration, we will not be amending our fact check.
Kind regards,
Bethan
October 7th 2021
Dear Bethan
Thank you for your reply.
You say your ‘fact check’ is related to our headline. Please can you spell out more precisely for me what you object to in the headline? Is it because it doesn’t include the word ‘unadjusted’ before ‘vaccine effectiveness’? Or is it something else? Unadjusted vaccine effectiveness is still a form of vaccine effectiveness so the headline is not inaccurate on that point (and the caveats are explained in the piece). Part of the problem is that you seem to regard vaccine effectiveness as something which can only be calculated in a formal study, rather than a quantity representing the reduced proportion of infections in a vaccinated group versus an unvaccinated group which may be calculated on any such data set (with limitations acknowledged). It is therefore not ‘incorrect’, as you claim, for me to calculate vaccine effectiveness from population data and report on it.
I appreciate that you are happy with the information you have included on vaccine effectiveness. However, the important point is it is not valid to claim that an article using more up-to-date data on real-world infection rates among vaccinated and unvaccinated groups is ‘incorrect’ by citing out-of-date estimates from studies using data from earlier periods, even if they come from government sources. You can point out that the new estimates disagree with the old estimates, but that doesn’t invalidate the new estimates or make them ‘incorrect’. What you are doing amounts to attempted censorship of reporting on emerging data, rather than ‘fact-checking’.
You say you disagree that you have misunderstood the PHE report. But you clearly imply that the PHE report does not show infection rates higher in the vaccinated than the unvaccinated. To quote:
This data had already caused widespread confusion, because it seemed to show for the month in question (August 9th to September 5th) that people in their 40s, 50s, 60s and 70s were more likely to test positive for Covid if they had been vaccinated than if they hadn’t. In particular, a chart displaying the data seemed to give this impression.
This is a patently misleading section as you completely fail to acknowledge that the report plainly does show infection rates higher in the vaccinated in these age groups and instead attempt to make it sound like it does not and that this was a matter of ‘confusion’ on the part of others. The PHE report even explicitly states: “In individuals aged 40 to 79, the rate of a positive COVID-19 test is higher in vaccinated individuals compared to unvaccinated.”
I urge you again, as a matter of professional integrity and for the sake of the credibility of your site, to amend the ‘fact check’ so that it is not misleading in this way and makes clear that the PHE report is correctly understood as showing infection rates higher in the vaccinated in these age groups during this time period.
Kind regards
Will
Will Jones
Associate Editor – Daily Sceptic
October 9, 2021
Posted by aletho |
Deception, Full Spectrum Dominance | COVID-19 Vaccine, UK |
Leave a comment
The University of California, Irvine has placed their Director of Medical Ethics, Dr. Aaron Kheriaty, on ‘investigatory leave’ after he challenged the constitutionality of the UC’s vaccine mandate in regards to individuals who have recovered from Covid and have naturally-acquired immunity.
Last month Kheriaty, also a Professor of Psychiatry at UCI School of Medicine, filed a suit in Federal court over the mandate.
“Natural immunity following Covid infection is equal to (indeed, superior to) vaccine-mediated immunity. Thus, forcing those with natural immunity to be vaccinated introduces unnecessary risks without commensurate benefits—either to individuals or to the population as a whole—and violates their equal protection rights guaranteed under the Constitution’s 14th Amendment,” Kheriaty wrote in a Sep. 21 blog post.
“Expert witness declarations in support of our case include, among others, a declaration from distinguished UC School of Medicine faculty members from infectious disease, microbiology/immunology, cardiology, endocrinology, pediatrics, OB/Gyn, and psychiatry,” the post continues (click here to read the rest).
…there is now considerable evidence that Covid recovered individuals may be at higher risk of vaccine adverse effects compared to those not previously infected (as seen in studies here, here, and here, among others). -Dr. Aaron Kheriaty
In a Wednesday update, Kheriaty writes that he’s been placed on ‘investigatory leave’ over his failure to comply with the mandate:
Via Human Flourishing :
Here is the latest move by the University of California in response to my lawsuit in Federal court challenging their vaccine mandate on behalf of Covid-recovered individuals with natural immunity. Last Thursday Sept 30th at 5:03 PM I received this letter from the University informing me that, as of the following morning, I was being placed on “Investigatory Leave” for my failure to comply with the vaccine mandate. I was given no opportunity to contact my patients, students, residents, or colleagues and let them know I would disappear for a month. Rather than waiting for the court to make a ruling on my case, the University has taken preemptive action:

You might be thinking, a month of paid leave doesn’t sound so bad. But the language is misleading here, since half of my income from the University comes from clinical revenues generated from seeing my patients, supervising resident clinics, and engaging in weekend and holiday on-call duties. So while on leave my salary is significantly cut. Furthermore, my contract stipulates that I am not able to conduct any patient care outside the University: to see my current patients, or to recoup my losses by moonlighting as a physician elsewhere, would violate the terms of my contract.
It came as no surprise that, since my request for a preliminary injunction was not granted by the court, the University would immediately begin procedures to dismiss me. However, in the complicated legal game of three-dimensional chess I did not anticipate this particular development: the current administrative designation, where I am neither able to work at the University nor permitted to pursue work elsewhere, was not a development I had anticipated. The University may be hoping this pressure will lead me to resign “voluntarily,” which would remove grounds for my lawsuit: if I resign prior to being terminated by the University, I have no legal claim of harm.
I have no intention at this time of resigning, withdrawing my lawsuit, or having an unnecessary medical intervention forced on me, in spite of these challenging circumstances. You may be wondering about the CA Department of Public Health vaccine mandate mentioned in the University’s letter above: yes, I am subject to two mandates, the UC mandate as a faculty member and the CA State mandate as a healthcare provider. Regarding the latter mandate, I filed a similar lawsuit in Federal court last Friday against the State Public Health Department. I will post more later on that case as it develops.
Although this is a challenging time for me and my family, at this time I remain convinced that this course of action is worthwhile. I am grateful for your ongoing encouragement, prayers, and support. I want my readers to know that am taking legal action not primarily for myself, but for all those who have no voice and whose Constitutional rights are being steamrolled by these mandates. As I wrote in my first post:
In my position, I came to see the importance of representing those whose voices were silenced, and to insist upon the right of informed consent and informed refusal. I have nothing personal to gain by this lawsuit and a lot to lose professionally. In the end, my decision to challenge these mandates came down to this question: How can I continue to call myself a medical ethicist if I fail to do what I am convinced is morally right under pressure?
Many of you have asked how you can support me and my efforts to challenge coercive mandates. My first answer is to consider becoming a paid subscriber to this newsletter if you are not already, and share this newsletter with others who are interested in following these issues. In the coming weeks I will be expanding my work on this Substack platform with live podcasts and audience Q&A for paid subscribers.
(Subscribe here)
For those who may wish to contribute more: I serve as Senior Fellow and Director of the Program in Health & Human Flourishing at the Zephyr Institute in Palo Alto, California. For the foreseeable future, the Program I direct there will focus on gathering and supporting experts, scholars, and leaders who are questioning various aspects of our response to this pandemic, and who are offering more effective solutions to the challenges we are facing. You can contribute to my work at the Zephyr Institute by making a donation HERE and specifying that you want your gift to support “Dr. Kheriaty’s work in the Health and Human Flourishing Program.”
This legal fight is important not only to set appropriate limits to vaccine mandates. It is also important for the future that—now in this crucial moment—we refuse to allow our institutions to set dangerous and unjust precedents. Today’s precedents could later facilitate even more coercive mandates and infringements on civil liberties by unelected officials, done during a declared “state of exception” or emergency that has no defined terminus—a dangerous precedent for a democratic society.
I want to thank all of you for being a part of this movement and for engaging with and encouraging my work on this issue. I could not do this without you.
* * *
Kheriaty’s situation is similar to that of Canadian ethics professor, Dr. Julie Ponesse, who made headlines last month after filming a now-viral tear-filled statement before she was fired by Huron University College in Ontario.
October 8, 2021
Posted by aletho |
Civil Liberties, Science and Pseudo-Science | COVID-19 Vaccine, United States |
Leave a comment

Before COVID-19, Moderna was in danger of hemorrhaging investors, as persistent safety concerns and other doubts about its mRNA delivery system threatened its entire product pipeline. Fear caused by the pandemic crisis made those concerns largely evaporate, even though there is no proof that they were ever resolved.
Those analyzing the COVID-19 crisis and its effects have mostly focused on how its disruptive nature has led to major shifts and recalibrations throughout society and the economy. Such disruption has also lent itself to a variety of agendas that had required an event of “reset” potential in order to be realized. In the case of the vaccine industry, COVID-19 has led to dramatic changes in how federal agencies manage the approval of medical countermeasures during a declared crisis, how trials for vaccine candidates are conducted, how the public perceives vaccination, and even how the term “vaccine” is defined.
Such shifts, though obvious, have provoked praise from some and sharp criticism from others, with the latter category being largely censored from public discourse on television, in print, and online. However, in objectively analyzing such seismic changes, it’s clear that most of these shifts in vaccine development and vaccine policy dramatically favor speed and the implementation of new and experimental technology at the expense of safety and thorough study. In the case of vaccines, it can be argued that no one benefitted more from these changes than the developers of the COVID-19 vaccines themselves, particularly the pharmaceutical and biotechnology company Moderna.
Not only did the COVID-19 crisis obliterate hurdles that had previously prevented Moderna from taking a single product to market, it also dramatically reversed the company’s fortunes. Indeed, from 2016 right up until the emergence of COVID-19, Moderna could barely hold it together, as it was shedding key executives, top talent, and major investors at an alarming rate. Essentially, Moderna’s promise of “revolutionizing” medicine and the remarkable salesmanship and fund-raising capabilities of the company’s top executive, Stéphane Bancel, were the main forces keeping it afloat. In the years leading up to the COVID-19 crisis, Moderna’s promises—despite Bancel’s efforts—rang increasingly hollow, as the company’s long-standing penchant for extreme secrecy meant that—despite nearly a decade in business—it had never been able to definitively prove that it could deliver the “revolution” it had continually assured investors was right around the corner.
This was compounded by major issues with patents held by a hostile competitor that threatened Moderna’s ability to turn a profit on anything it might manage to take to market, as well as major issues with its mRNA delivery system that led them to abandon any treatment that would require more than one dose because of toxicity concerns. The latter issue, though largely forgotten and/or ignored by media today, should be a major topic in the COVID-19 booster debate, given that there is still no evidence that Moderna ever resolved the toxicity issue that arose in multi-dose products.
In this first installment of a two-part series, the dire situation in which Moderna found itself immediately prior to the emergence of COVID-19 is discussed in detail, revealing that Moderna—very much like the now disgraced company Theranos—had long been a house of cards with sky-high valuations completely disconnected from reality. Part 2 will explore how that reality would have come crashing down sometime in 2020 or 2021 were it not for the advent of the COVID-19 crisis and Moderna’s subsequent partnership with the US government and the highly unusual processes involving its vaccine’s development and approval. Despite the emergence of real-world data challenging the claims that Moderna’s COVID-19 vaccine is safe and effective, Moderna’s booster is being rushed through by some governments, while others have recently banned the vaccine’s use in young adults and teens due to safety concerns.
As this two-part series will show, safety concerns about Moderna were known well before the COVID crisis, yet they have been ignored by health authorities and the media during the crisis itself. In addition, in order to stave off collapse, Moderna must keep selling its COVID-19 vaccine for years to come. In other words, without the approval of its booster, which has caused great controversy even among the country’s top vaccine officials, Moderna faces a massive financial reckoning. While the COVID-19 crisis threw the company a lifeboat, the administration of its COVID-19 vaccine, in which the US government has now invested nearly $6 billion, must continue into the foreseeable future for the bailout to be truly successful. Otherwise, a company now worth $126.7 billion, with major investments from the US government, US military, and ties to the world’s wealthiest individuals, will crumble in short order.
A New Theranos?
In September 2016, Damian Garde, the national biotech reporter for the medical media company STAT, wrote a lengthy exposé of the “ego, ambition, and turmoil” plaguing “one of biotech’s most secretive startups.” The article focused on the company Moderna, which had been founded in 2010 to commercialize the research of Boston Children’s Hospital cell biologist Derrick Rossi. The effort to turn a profit by creating Moderna, which intimately involved controversial scientist and close Bill Gates associate Bob Langer as well as Cambridge, Massachusetts–based Flagship Ventures (now Flagship Pioneering), began soon after Rossi published a report on the ability of modified RNA to turn skin cells into different types of tissue.
Between the time of Moderna’s founding and Garde’s 2016 investigation, the buzz around Rossi’s research and its potential to create medical breakthroughs had waned, as had the buzz around its potential to make its investors very wealthy. Despite teaming up with pharmaceutical giants like AstraZeneca and raising record amounts of funding, Moderna still had no product on the market six years after its founding, and, as STAT revealed, the “company’s caustic work environment” had led to a persistent hemorrhaging of top talent, though little of its internal conflicts was publicly known due to “its obsession with secrecy.” Most troubling for the company that year, however, was that Moderna appeared to have “run into roadblocks with its most ambitious projects.”

Aside from the scientific obstacles that Moderna had encountered, one major “roadblock” for the company, per Garde, was none other than Stéphane Bancel, Moderna’s top executive, who still heads the company. According to Garde, Bancel was squarely at the center of many of the company’s controversies due, in part, to his “unwavering belief that Moderna’s science will work—and that employees who don’t ‘live the mission’ have no place in the company.” Between 2012 and 2016, Bancel was allegedly a key factor in the resignation of at least a dozen “highly placed executives,” including those who directed Moderna’s product pipeline as well as its vaccine projects.
Bancel, prior to joining Moderna, had spent much of his career in sales and operations, not science, making a name for himself at pharmaceutical giant Eli Lilly before heading a French diagnostics firm called bioMérieux. His performance there, as well as his ambition, caught the attention of Flagship Ventures, a Moderna cofounder and top investor, which then connected him with the company he would go on to lead.
Although lacking a background in mRNA and the science behind its use as a therapeutic, Bancel has made up for it by becoming Moderna’s salesman par excellence. Under his leadership, Moderna became “loath to publish its work in Science or Nature, but enthusiastic to herald its potential on CNBC and CNN.” In other words, under Bancel, the company came to promote its science through media publicity and public relations rather than by publishing actual data or scientific evidence. When two of its vaccine candidates entered phase 1 human trials in 2016 (trials that ultimately went nowhere), the company declined to list them on the public federal registry ClinicalTrials.gov. The decision not to list, which deviates from common practice by Moderna’s competitors and other more traditional vaccine companies, meant that the information on the safety of these vaccine candidates would likely never be publicly available after the trial’s conclusion. Moderna also refused to publicly comment on what diseases these vaccines were meant to target.
Such secrecy became commonplace at Moderna after Bancel took the helm, with the company having published no data “supporting its vaunted technology” by the time STAT’s 2016 exposé was published. Insiders as well as investors that had committed millions to the company were only granted “a peek” at the company’s data. According to former Moderna scientists who spoke to STAT, the company was “a case of the emperor’s new clothes.” Former employees further charged that Bancel was actually “running an investment firm” and “then hop[ing] it also develops a drug that’s successful.”
Perhaps this is why Bancel was deemed the best executive to steer Moderna. As an ambitious salesman running a highly overvalued company, he would prioritize the company’s image and its finances regardless of any issues with the science underpinning it all. Perhaps it was for that reason that Bancel, per former employees, “made it clear [from the beginning] that Moderna’s science simply had to work. And that anyone who couldn’t make it work didn’t belong.”
As STAT noted in 2016, the people who were tasked with making “the science work” were those who most frequently resigned, which led to Moderna losing two heads of chemistry within a single year, followed shortly by losing its chief scientific officer and its head of manufacturing. Many top executives, including the heads of its cancer research and rare disease research branches, ended up lasting fewer than eighteen months in their respective positions. The abrupt resignations weren’t exclusive to Moderna’s science-focused executive positions either, as the chief information officer and top financial executive role were also affected. Bancel ultimately sought advice from the human resources departments of Facebook, Google, and Netflix on employee retention.
Particularly telling was the abrupt and mysterious resignation of Moderna’s head of research and development, Joseph Bolen, after about two years at the company. A company insider at the time told STAT that the only reason Bolen would have resigned was if “there was something wrong with the science or the personnel.” In other words, Bolen either left because the science underpinning Moderna’s massive valuation did not live up to the hype or Bancel had forced him out, with the additional possibility that both were key in Bolen’s resignation.
Speculation at the time pointed the finger at Bancel, though it’s not clear why the rift between the two men emerged. Bancel asserted that he tried to convince Bolen to stay, though there were contrasting assertions from anonymous employees, and that Bolen had “voted himself off the island.”
Whatever the exact cause of the resignation of the head of R & D, it only added to the mystique around Moderna’s inner workings and its ability to deliver on its promise to “revolutionize” medicine. It also reveals more than a few similarities between Moderna and the now-disgraced company Theranos. Theranos, whose former top executive, Elizabeth Holmes, is now on trial for fraud, was known for its extreme culture of secrecy that kept investors and business partners in the dark, forced nondisclosure agreements on everyone who came in contact with the company, and kept employees “siloed” through an extremely strict need-to-know policy. Like Moderna, Theranos had been praised as revolutionary and poised to “change the medical industry forever.” Similarly, its top executive had no professional health-care or science experience, yet both fired or forced the resignations of employees who disagreed with their perspective or were unable to provide “positive” results. Both companies also failed to publish any evidence in peer-reviewed journals that the science behind their multibillion-dollar valued companies was more than just fantasy and a well-devised sales pitch.
Arguably, the most critical difference between Moderna and Theranos is that Moderna, whose numerous issues and challenges only came to light after the collapse of Theranos had begun, has never faced the same degree of scrutiny from the US government or mainstream investigative journalists. There are many possible reasons for this, including Moderna’s close relationship with the US Department of Defense through the Defense Advanced Research Projects Agency (DARPA), or concern that its exposure post-Theranos would bring scrutiny to any company existing at the intersection of Silicon Valley and the health-care industry. However, such a reckoning would likely have been inevitable for Moderna had it not been for the COVID-19 crisis, which could not have come at a more convenient time for the company.
Moderna’s “Software” Encounters Bugs
Many of the problems with Moderna that Garde identified in 2016 continued to plague the company right up until the beginning of the COVID-19 crisis. Chief among these was Moderna’s struggle to prove that its technology worked and that it was safe. Concerns about the safety and efficacy of the company’s products, which were publicly reported beginning in 2017, evaporated in the wave of panic surrounding COVID-19 and the simultaneous “Warp Speed” race for a vaccine that would “end the pandemic.” Yet, there is little, if any, evidence that these once-well-recognized concerns were addressed prior to the US government’s emergency use authorization of Moderna’s COVID-19 vaccine and its now widespread use in many countries around the world. To the contrary, there is evidence that these concerns were covered up both prior to and during the development of its vaccine.

The reports that emerged in January 2017 noted that Moderna had “run into troubling safety problems with its most ambitious therapy” and that the company was “now banking on a mysterious new technology to keep afloat.” The “ambitious therapy” in question was meant to treat Crigler-Najjar syndrome and “was to be the first therapy using audacious new technology that Bancel promised would yield dozens of drugs in the coming decade.” Bancel had specifically used the Crigler-Najjar therapy as a major selling point to investors, particularly in 2016 when he touted it at the JP Morgan Healthcare Conference.
Yet, employees of Alexion, the company co-developing the drug with Moderna, blew the whistle on the project in 2017, revealing that it “never proved safe enough to test in humans” and that the failure of this therapy and the technology platform it sought to use had been responsible for prompting Moderna to abandon the class of drug therapies that, for years, had justified its sky-high valuation and attracted hundreds of millions in investor cash.
As a result of the problem with the Crigler-Najjar drug, media outlets asserted that Moderna was now “in need of a Hail Mary” that would keep its valuation from imploding and its investors from fleeing. The persistence of problems first noted in the 2016 STAT investigation, such as Moderna’s failure to publish meaningful data supporting its mRNA technology, were only exacerbating the company’s increasingly precarious position. Indeed, not long before the indefinite delay of the Crigler-Najjar therapy, Bancel had dismissed questions about Moderna’s promise by painting mRNA as an easy way to quickly develop novel treatments for a variety of diseases. He stated that “mRNA is like software: You can just turn the crank and get a lot of products going into development.” If that were the case, why did the company have no products on the market after nearly seven years, and why had its most touted project experienced such obstacles? Clearly, in keeping with Bancel’s “software” metaphor, Moderna’s technology had encountered bugs, bugs that were potentially ineradicable.
It turns out that the Crigler-Najjar drug therapy that Moderna had bet on so heavily had failed because of the lipid nanoparticle delivery system it used to transport mRNA into cells. Crigler-Najjar had been chosen as a target condition because Moderna scientists deemed it to be “the lowest-hanging fruit.” First, the syndrome is caused by one specific genetic defect; second, the affected organ, the liver, is among the easiest to target with nanoparticles; and third and most important for the company, treating the disease with mRNA would require frequent doses, ensuring a steady stream of income for the company. Thus, given the first two motives behind the company’s focus on Crigler-Najjar, if Moderna couldn’t develop a therapy for that condition, it meant they wouldn’t be able to develop a therapy for other conditions that, for example, were caused by multiple genetic defects or affected multiple organs or those more resistant to nanoparticle-based treatments. In other words, that “Moderna could not make its therapy [for Crigler-Najjar] work” meant that it was unlikely to make therapies of that entire class work either.
Indeed, media reports on the indefinite delay of this particular therapy noted that “the indefinite delay on the [Moderna] Crigler-Najjar project signals persistent and troubling safety concerns for any mRNA treatment that needs to be delivered in multiple doses.” This issue would soon lead Moderna to only pursue treatments that could be delivered as a single dose—that is, until the emergence of COVID-19 and the advent of the COVID-19 vaccine booster debate. It is also worth mentioning that, due to the extreme rarity of Crigler-Najjar syndrome, even if the therapy had been successfully taken to market by Moderna, it would have been unlikely to bring in enough money to sustain the company.
The specific problem Moderna encountered with the Crigler-Najjar treatment was related to the lipid nanoparticle delivery system it was using. According to former Moderna employees and their collaborators at Alexion, “The safe dose was too weak, and repeat injections of a dose strong enough to be effective had troubling effects on the liver [the target organ of this particular therapy] in animal studies.” This was an issue Moderna had apparently run into with its nanoparticle delivery system in other cases too, according to reports published at the time. Per STAT, the delivery system employed by Moderna had consistently “created a daunting challenge: Dose too little, and you don’t get enough enzyme to affect the disease; dose too much, and the drug is too toxic for patients.”
Moderna attempted to offset the bad press over having to delay the Crigler-Najjar drug with claims that they had developed a new nanoparticle delivery system called V1GL that “will more safely deliver mRNA.” The claims came a month after Bancel had touted another delivery system called N1GL to Forbes. In that interview, Bancel told Forbes that the delivery system they had been using, licensed to them by Acuitas, “was not very good” and that Moderna had “stopped using Acuitas tech for new drugs.” However, as will be explored in detail in this report as well as Part II of this series, it appears that Moderna continued to rely on the Acuitas-licensed technology in subsequent vaccines and other projects, including its COVID-19 vaccine.
Former Moderna employees and those close to their product development were doubtful at the time that these new and supposedly safer nanoparticle delivery systems were of any consequence. According to three former employees and collaborators close to the process who spoke anonymously to STAT, Moderna had long been “toiling away on new delivery technologies in hopes of hitting on something safer than what it had.” All of those interviewed believed that “N1GL and V1GL are either very recent discoveries, just in the earliest stages of testing—or else new names slapped on technologies Moderna has owned for years.” All spoke anonymously due to having signed nondisclosure agreements with the company, agreements that are aggressively enforced.
One former employee, commenting on the alleged promise of N1GL and V1GL, stated that these platforms “would have to be a miraculous, Hail Mary sort of save for them to get to where they need to be on their timelines. . . . Either [Bancel] is extremely confident that it’s going to work, or he’s getting kind of jittery that, with a lack of progress, he needs to put something out there.”

Stephen Hoge, Moderna’s president, and Melissa Moore, Moderna’s CSO for Platform Research Source: Moderna
It seems that those former employees who believed that N1GL and V1GL were new names put on existing technology and that Bancel was overselling their promise were correct, as Moderna appears to have returned to the troubled lipid nanoparticle delivery system it had licensed from Acuitas for subsequent therapies, including its COVID-19 vaccine. As will be explored in this report and Part II of this series, there is no evidence that Moderna ever got their “Hail Mary” save when it came to acquiring the rights for or developing a safe mRNA delivery system.
On top of the much-touted promises of N1GL and V1GL as safer treatments, Moderna additionally vowed to create “new and better formulations” for the Crigler-Najjar therapy that could potentially make it to human trials at a later time. This helped to stave off more bad press, but only for a few weeks. One month after the troubles with the Crigler-Najjar therapy were publicly reported, the head of Moderna’s oncology division, Stephen Kesley, left the company. This was just as Moderna was moving toward its first human trials for its cancer treatment, which forced “a senior leadership team with little experience in developing drugs to sort out the company’s future in the field.” Just weeks before Kesley’s departure, Bancel had boldly claimed in a bid to woo new investors at the JP Morgan Healthcare Conference, held in January 2017 in San Francisco, that oncology was Moderna’s “next big opportunity after vaccines.”
The same month as Kesley’s departure, Moderna was able to draw media attention elsewhere, as for the very first time they published data in a peer-reviewed journal. In Cell, its scientists published data on an animal trial for its Zika vaccine candidate that positively demonstrated both efficacy and safety in mice. While animal trial results do not necessarily translate into equivalent results in humans, the results were deemed to “bode well” for Moderna’s planned clinical trial of that vaccine candidate in humans. In addition, the results were like the animal trial results published by Moderna competitor BioNTech for their mRNA vaccine candidate for Zika a month earlier.
However, for Moderna, the positive news was muted by a negative ruling on a legal dispute that threatened Moderna’s ability to ever turn a profit on the Zika vaccine or any other mRNA vaccine it developed, a threat that Moderna’s competitors, such as BioNTech, didn’t have to contend with. That ruling, discussed in greater detail later in this report, greatly restricted Moderna’s use of the lipid nanoparticle delivery system licensed to it through Acuitas and directly threatened the company’s ability to create a for-profit product using intellectual property tied to the relevant patents. It would also kick off a years-long legal dispute that has suggested at various times that the promises of V1GL and N1GL were either completely invented or greatly exaggerated, as former Moderna employees and collaborators had stated.
Not long afterward, in July 2017, Moderna was hit with yet another wave of bad press as their partner in the Crigler-Najjar venture, Alexion, cut ties with the company completely. Moderna downplayed Alexion’s decision and claimed it had acquired “extensive knowledge” that would allow it to continue to develop the troubled therapy on its own. Nonetheless, Alexion’s decision came at an inopportune time for the company, as one of Moderna’s top investors had just two weeks earlier slashed its valuation of the company by almost $2 billion, allegedly because Moderna had “struggled to live up to its own hype.” Reports began to circulate claiming that “Moderna’s investors might be losing faith in the company’s future.”
Indeed, the Crigler-Najjar syndrome drug was not the only one that, at that point, had proven “too weak or too dangerous to test in clinical trials,” according to former employees and partners. The persistent issue, which again lay with the nanoparticle delivery system Moderna had licensed from Acuitas, had forced the company, beginning with the delay of the Crigler-Najjar therapy, to “prioritize vaccines, which can be dosed just once and thus avoid the safety problems that have plagued more ambitious projects.”
Yet, these single-dose “vaccines” or therapies were considered not as lucrative as the drug therapies Moderna had long promised and that underpinned its multibillion-dollar valuation, thereby forcing the company to “bet big on a loss-leader.” Also problematic was that Moderna lagged behind its mRNA vaccine competitors and that the supposed promise of its technology to produce viable vaccines was only “proven” at that point by a single, small trial. That trial, as noted by the Boston Business Journal, was an “early-stage human trial that was primarily meant to assess the safety of an avian flu vaccine.” Moderna had claimed, despite the trial being designed to assess safety, that it had “provided evidence that the vaccine is effective, with no major side effects” as well. Furthermore, as will be discussed in a later section of this report, the legal dispute over the Acuitas-licensed lipid nanoparticle system threatened Moderna’s ability to ever turn on a profit on any mRNA vaccine it managed to get through trials and the federal approval process, making the company’s future appear quite grim.
Despite Positive Press, Lingering Questions Remained
In September 2017, at a closed-door investor event meant to prevent more major investors from devaluing the company or jumping ship, Moderna provided more insight into a recently published press release on the trial results of a therapy meant to regrow heart tissue by boosting production of a protein known as VEGF. The press release, which generated positive media headlines, noted that the therapy had been proven safe in a study with a sample size of 44 patients. However, neither the press release nor the data Moderna disclosed to investors at the closed-door meeting revealed how much protein the therapy caused patients to produce, leaving its efficacy a mystery. Indeed, media reports on the investor meeting noted that “since Moderna did not release that crucial data point, outsiders can’t judge how much therapeutic potential there may be.”
The results, though they seemed to mitigate the concerns over the safety of Moderna’s technology, failed to inspire confidence in many attendees. Several attendees later told reporters that they “were not overly impressed” with Moderna’s presentation, which only “underlined lingering questions about whether it can live up to its own hype.”
One of the issues here, yet again, is that Moderna’s valuation was and is underpinned by its promise to produce products for rare diseases that require repeated injections over a patient’s lifetime. The VEGF therapy promoted by Moderna at this meeting was meant to be a one-time-only injection, and, thus, evidence of its safety did not resolve the problem of none of Moderna’s multi-dose products having proven safe enough to test on humans. The closed-door investor event made it clear that Moderna was aiming to avoid that persistent problem by prioritizing single-dose vaccines.
As STAT noted at the time:
The presentation to investors also made clear that Moderna is prioritizing vaccines. They are easier to develop from mRNA because patients need just one dose, which eliminates some of the safety issues that have plagued more ambitious projects such as therapies for rare diseases.
The pivot to vaccines remained a sore point with many investors, however, as vaccines are viewed as “low-margin product[s] that can’t generate anywhere near the profits seen in more lucrative fields like rare diseases and oncology.” These, as previously mentioned, are the very fields on which Moderna’s massive valuation had been based but for which it had been unable to produce safe and effective therapies. Moderna was clearly aware of these concerns among its current and potential investor base and attempted to speak promisingly of its oncology-related efforts at this same event. However, it was silent on trial timing and other key data points, maintaining the company’s long-standing reputation for secrecy towards both insiders and the general public. It is certainly telling that Moderna remained so secretive about key data at an event not only closed to the public and the press, but meant to reassure existing investors and to entice new ones. If Moderna declined to show important data to investors at a time when it was desperately seeking to keep them onboard, it implies that the company either had something to hide or nothing to show.
Moderna’s increasingly troubled internal situation, despite its consistently rosy PR, escalated a month later when reports emerged of the abrupt resignations of its head of chemistry, the leader of its cardiovascular division, and the head of its rare diseases division. These resignations, which occurred toward the end of 2017, followed the high-profile resignations the company suffered that were mentioned in the 2016 STAT exposé by Damian Garde.
A few months later, in March 2018, the chief scientific officer of Moderna’s vaccine business, Giuseppe Ciaramella, also left. This resignation signaled further internal troubles at the company, even more because Moderna had recently and very publicly pivoted to vaccines; and Ciaramella, in addition to leading vaccine development at this critical juncture, had been the first Moderna executive to suggest that the company’s technology could be useful in developing vaccines, a suggestion that the company was now betting everything on. One can’t help but wonder if Bancel’s tendency to force out employees and executives who “couldn’t make the science work” was a factor in any of these high-profile resignations, including that of Ciaramella.
A Years-Long Legal Snafu
Thus far, this report has largely focused on how Moderna’s extreme secrecy appears to have been used to obfuscate and mitigate major issues with its technology and product pipeline and how those issues were reaching a climax following the company’s IPO and immediately prior to the COVID crisis. However, the challenge of creating products that work and can be proven to work in clinical settings is but one of at least two major issues facing Moderna as a company. Indeed, during the same timetable explored above, Moderna was embroiled in aggressive disputes related to intellectual property and patents. Notably, these same legal issues deal with the lipid nanoparticle system that was also reportedly at the root of Moderna’s safety and product-pipeline issues.
As mentioned earlier, the lipid nanoparticle delivery system used in many Moderna therapies was licensed to them by Acuitas. Acuitas, however, had licensed that system from a separate company, Arbutus, which sued in 2016 claiming that Acuitas’s sublicense to Moderna was illegal. Arbutus won the case, which lead to a temporary injunction in 2017 that stopped Acuitas from further sublicensing the lipid nanoparticle technology. A settlement reached between Acuitas and Arbutus in 2018 terminated Acuitas’s license and restricted Moderna’s use of the technology to four vaccine candidates that targeted already identified viruses.
Moderna’s Bancel told Forbes in 2017 that the Acuitas/Arbutus system was barely mediocre and that Moderna was developing its own improved delivery system that would not infringe on Arbutus’s intellectual property (the aforementioned N1GL and V1GL systems). However, soon after Bancel made those claims, Arbutus’s leadership challenged them, stating that the company had reviewed all of Moderna’s patents, publications, and presentations regarding these “new” delivery systems and had found nothing that didn’t involve their own intellectual property. Even former Moderna employees, as mentioned previously, were very doubtful that N1GL and V1GL were any different than the Acuitas/Arbutus system, meaning that—despite Bancel’s claims—Moderna had unresolved legal woes related to these nanoparticles that, along with the toxicity issues, was stalling Moderna product candidates.
It is important to note at this point that, while only Moderna has been locked in a legal battle with Acuitas/Arbutus for years over LNP intellectual property, the other main producers of mRNA COVID-19 vaccines, Pfizer/BioNTech and CureVac, also use major aspects of the same Arbutus-derived technology. However, BioNTech licensed the LNPs in such a way as to avoid the issues that have entangled Moderna for years.
Moderna’s legal dispute, in addition to the already discussed safety issues, greatly threatened Moderna’s ability to survive as a company. Having already been forced to settle on the vaccines market and reject the more lucrative and “revolutionary” mRNA therapies it had long promised, Moderna was steadily moving toward a position where it had “no right to sell” vaccine products that depended on the Arbutus-patented and Acuitas-sublicensed technology. This situation has placed pressure on Moderna to negotiate a new license with Arbutus directly, negotiations in which the company would have very little leverage.
Since the first legal case in 2016, Moderna and Arbutus have remained locked in disputes about the nanoparticles and who owns them. Moderna challenged three Arbutus patents with the US Patent and Trademark Office, with mixed results. Yet, simultaneously, Moderna also claimed that its tech was “not covered by the Arbutus patents,” which prompted numerous observers and reporters to ask questions such as—“In that case, why did [Moderna] initiate the legal action against Arbutus to begin with?”
Moderna answered that query by claiming that it targeted Arbutus only because of Arbutus’s past “aggression” against them. However, despite such claims, the effort and cost inherent in the legal challenge reveals that, at the very least, Moderna takes the threat of Arbutus’s intellectual property claims very seriously. The actual answer seems to lie in Moderna being willing to publicly claim that their LNP technology is different enough from the Arbutus-derived system covered by the patents but unwilling to release any proof—whether in court, to its own investors, or to the public—that it is in fact different. The more recent twists and turns of this protracted legal battle, including a pivotal 2020 decision that was very unfavorable for Moderna, are discussed in Part II of this series.
Anything to Aid a Slumping Stock Price

Nasdaq building on the day of Moderna’s 2018 IPO. Source: Nasdaq
Just previous to Ciaramella’s resignation, Moderna had claimed to have “solved the scientific issues that made its earlier mRNA treatments too toxic for clinical trials,” according to media reports. Those reports also claimed that, as a result, “Moderna believes it has steered back on course,” though the company did not provide evidence to support that claim. Nevertheless, the promise allowed the company to complete a new round of financing, during which it raised an additional $500 million from “an investor syndicate uncommon in biotech” that included the governments of Singapore and the United Arab Emirates. Some observers were puzzled as to how Moderna had managed to raise so much money despite the outstanding questions about the science underpinning its high valuation.
The answer came with the publication of Moderna’s confidential investor slide deck by STAT’s Damian Garde, which showed that the company had predicted that drugs that they had only been tested in mice would soon be worth billions and that its vaccine revenue would amount to $15 billion annually. The slide deck, deemed “pretty absurd” and “geared at hopeful generalists that can dream big” per one skeptical investor, made it clear why the company’s last funding round had appealed to “unconventional” biotech investors rather than veteran investors focused on the industry. A veteran biotech investor, who spoke anonymously due to the slide deck’s confidentiality, stated that “it’s a deck designed to tell the ‘we’re going to be huge’ story to a group of rather unsophisticated investors—and it does that beautifully. . . Just enough science and platform stuff to convey the ‘We know what we’re doing’ sentiment, but not enough to engender technical questions.”
Moderna slide deckDownload
Per those who sat through Moderna’s pitch, the company was “very generous on the market-size assumptions for their programs,” with one former Moderna collaborator placing the real-world value of a treatment the company had claimed was worth billions annually at closer to “$100 million to 250 million.” Of course, that revenue estimate comes with the caveat that the treatment, tested thus far only in mice, would someday prove to work in humans. A former Moderna employee in its rare diseases division stated at the time that Moderna “continue[s] to rush forward and over-promise the potential for broad use of mRNA prior to any evidence beyond vaccines or very early experiments in mice.”
Despite Moderna’s ability to convince “unsophisticated” and/or “unconventional” investors to back its early 2018 funding round, it appears that one of its most important promises used to attract investors—that it had solved the nanolipid particle toxicity issue—was not true.
In a filing with the Securities and Exchange Commission dated November 2018, months after Moderna had claimed to have fixed the issues with its lipid nanoparticle delivery system, the company made several claims that appear to contradict its purported development of a new, safer nanoparticle technology.
For example, the filing states on page 33:
Most of our investigational medicines are formulated and administered in an LNP [lipid nanoparticle] which may lead to systemic side effects related to the components of the LNP which may not have ever been tested in humans. While we have continued to optimize our LNPs, there can be no assurance that our LNPs will not have undesired effects. Our LNPs could contribute, in whole or in part, to one or more of the following: immune reactions, infusion reactions, complement reactions, opsonation [sic] reactions, antibody reactions including IgA, IgM, IgE or IgG or some combination thereof, or reactions to the PEG from some lipids or PEG otherwise associated with the LNP.
Certain aspects of our investigational medicines may induce immune reactions from either the mRNA or the lipid as well as adverse reactions within liver pathways or degradation of the mRNA or the LNP, any of which could lead to significant adverse events in one or more of our clinical trials. Many of these types of side effects have been seen for legacy LNPs. There may be resulting uncertainty as to the underlying cause of any such adverse event, which would make it difficult to accurately predict side effects in future clinical trials and would result in significant delays in our programs. (emphasis added)
Based on these statements, Moderna appeared to be uncertain as to whether its current lipid nanoparticle delivery system was any safer than that which led to the indefinite delay of its Crigler-Najjar therapy. In addition, the reference to “adverse reactions within liver pathways,” one of the main issues that triggered the specific delay of the Crigler-Najjar therapy, suggests a continued reliance on technology sublicensed from Acuitas. As will be noted in Part II, the Moderna COVID-19 vaccine also appears to use the controversial Acuitas technology that had prompted significant safety, legal, and financial concerns for Moderna for years.
The November 2018 SEC filing makes other statements regarding its supposedly fixed lipid nanoparticle delivery system that are worth noting:
If significant adverse events or other side effects are observed in any of our current or future clinical trials, we may have difficulty recruiting trial participants to any of our clinical trials, trial participants may withdraw from trials, or we may be required to abandon the trials or our development efforts of one or more development candidates or investigational medicines altogether. . . .
Even if the side effects do not preclude the drug from obtaining or maintaining marketing approval, unfavorable benefit risk ratio may inhibit market acceptance of the approved product due to its tolerability versus other therapies. Any of these developments could materially harm our business, financial condition, and prospects.
These statements are significant in that they openly suggest at least one reason for Moderna’s long-standing tendency toward secrecy in publishing data about its treatments, as public knowledge of its technology’s persistent challenges would threaten its ability to attract trial participants, investors, and, later, consumers.
About a month after these troubling admissions were made in fine print, Moderna succeeded in pulling off a record-setting initial public offering (IPO) in December 2018. For that IPO, Moderna had retained the services of eleven investment banks, which is reportedly around “twice the number normally seen in biotech offerings.” However, its stock value tumbled just hours afterward, “a sign the company and its underwriters might have over-estimated demand for the richly valued company.” A month after the IPO, Moderna’s stock continued its downward slide, “doing exactly the opposite of what private investors look for in an IPO.” Those who had predicted this post-IPO outcome before Moderna went public had also warned that this downward trend would likely continue through early 2020 if not longer. Skeptics such as STAT’s Damian Garde had warned right before Moderna’s IPO that that the company’s sliding stock value would likely continue throughout 2019 due to “a seeming lack of impending news,” given that “momentum in biotech, positive or negative, is driven by catalysts” and “Moderna is in for a fairly quiet 2019.”
Meanwhile, media reports warned, as they had for years, that Moderna “is still in the early days of proving [their] technology’s potential,” despite being a nine-year-old company. Such reports also noted that Moderna’s inability to prove its technology’s worth after nearly a decade in business was hampered by its “struggl[e] in its initial efforts to turn mRNA into drugs that can be repeatedly dosed, leading it to pivot to vaccines, which can be administered just once or twice.” Investors at the 2019 JP Morgan health-care conference spoke of concerns that “Moderna [has] yet to rule out the lingering risks tied to mRNA, and, even at its depressed valuation, the company is simply too expensive.” Others confided in reporters that they would be “sitting on the sidelines until Moderna either changes the narrative with promising human data or gets substantially cheaper.”
A few weeks later, Moderna’s Bancel attended the World Economic Forum’s 2019 annual meeting alongside Johnson & Johnson executive Paul Stoffels and other pharmaceutical and biotech leaders in order to “rub elbows with world leaders and one-percenters—and talk about the future of healthcare.” Other health-care figures in attendance included head of the World Health Organization, Tedros Adhanom Ghebreyesus, and “global health philanthropist” Bill Gates, whose foundation entered into “a global health project framework” with Moderna in 2016 to “advance mRNA-based development projects for various infectious diseases.” The Bill & Melinda Gates Foundation is the only foundation listed as a “strategic collaborator” on the Moderna website. Other “strategic collaborators” include the US government’s Biomedical Advanced Research and Development Authority (BARDA), the US military’s DARPA, and pharmaceutical giants AstraZeneca and Merck.
Moderna first teamed up with the WEF just a few years after its founding back in 2013, when it was named to the Forum’s community of Global Growth Companies (GGC). That year, Moderna was one of just three North American health companies to receive the honor and was additionally recognized by the Forum as “an industry leader in innovative mRNA therapeutics.” “We are honored to be recognized for our efforts to advance our platform and ensure its potential is realized on a global scale, and we look forward to being a member of the World Economic Forum community,” Bancel said at the time.

Stéphane Bancel at the World Economic Forum Annual Meeting, January 2020. Source: WEF
As a WEF Global Growth Company, Moderna has closely and regularly engaged with the Forum since 2013 at both the Chinese-hosted Annual Meeting of the New Champions and the WEF’s regional meetings, while also having access to the WEF’s exclusive networking platform that provides the company privileged access to the world’s most powerful business and government leaders. Additionally, such carefully selected companies are given opportunities by the Forum “to shape global, regional and industry agendas and engage in meaningful exchanges about ways to continue on a sustainable and responsible path of growth.” Essentially, the roster of such companies constitutes a consortium of corporations that are promoted and guided by the Forum because of their commitment to “improving the state of the world,” that is, their commitment to supporting the Forum’s long-term agendas for the global economy and for global governance.
In April 2019, Moderna published some information on modifications to its lipid nanoparticles (discussed in more detail in Part II). A month later, in May 2019, Moderna published positive results in the journal Vaccine for phase 1 data on mRNA vaccine candidates for “two potential pandemic influenza strains” administered as two doses three weeks apart. The company’s press release on the study stated that “future development of Moderna’s pandemic influenza program is contingent on government or other grant funding,” suggesting that it would use the trial results to lobby the government for funds for a continuation of this particular program.
Notably, at the same time as these results were published, the US Department of Health and Human Services Office of the Assistant Secretary for Preparedness and Response, then filled by Robert Kadlec, was in the midst of conducting Crimson Contagion, a multimonth simulation of a global pandemic involving an influenza strain that originates in China and spreads globally through air travel. The strain at the center of the simulation, called H7N9, is one of the very strains used in the Moderna study. Moderna published those results on May 10, just four days before the Crimson Contagion simulation hosted its federal interagency seminar. BARDA, which the ASPR office oversees, is a major strategic ally of Moderna and was co-developing these “potential pandemic influenza” vaccines that are mentioned in this timely press release, that is, for H10N8 and H7N9 influenza infections.
Crimson Contagion is notable for several reasons, most significantly for Kadlec’s own history with the Dark Winter simulations that preceded and eerily predicted the 2001 anthrax attacks. As has been discussed in detail in a previous TLAV – Unlimited Hangout investigation, the 2001 anthrax attacks conveniently rescued anthrax vaccine manufacturer BioPort, now Emergent Biosolutions, from certain ruin, much like the way the COVID crisis did for Moderna.
A month later, in June 2019, Moderna again managed to generate positive headlines on making its debut at the American Society of Clinical Oncology annual meeting, where it sought to promote its ability to produce the personalized cancer treatments that had been key to wooing investors both before and after its record-setting IPO. It was the first time the company had publicly presented data on a cancer treatment, and this particular treatment was being co-developed with Merck. The data showed positive results in preventing relapses in cancer patients whose solid tumors had been removed via surgery, but the trial failed to show any definitive effect in cancer patients whose tumors had not been removed. Thus, the early data seemed to indicate that Moderna’s treatment would only help cancer patients stay in remission after other medical interventions had been performed. Though the news allowed Moderna to bask in some much-needed positive press and to promote its oncology products in development, some reports rightly noted that it was “still too early for any definitive judgment” on the cancer treatment’s clinical benefit.
Despite this apparent advance, by September 2019, Moderna’s stock price continued to decline, leading to a loss of about $2 billion in market value from the company’s $7.5 billion valuation at the time of its record-setting IPO. The main factors for this were the same persistent problems the company had been facing for years—lack of progress, including lack of products on the market; persistent safety problems with its mRNA technology; and the lack of data showing that advances were being made to make that technology commercially feasible.
In mid-September 2019, Moderna gathered investors together to showcase scientific evidence it claimed would finally prove that its mRNA technology could “turn the body’s own cells into medicine-making factories” and hopefully “turn skeptical investors into believers.” This data, which was derived from a very preliminary study that involved only four healthy participants, had complications. Three of the four participants had side effects that prompted Moderna to state at the meeting that they would need to reformulate the mRNA treatment to include steroids, while one of the participants suffered heart-related side effects, including a rapid heart rate and an irregular heartbeat. Moderna, which asserted that none of the heart-related side effects was serious, could not “definitively pinpoint the cause of the heart symptoms.” Yet, as previously mentioned, it was likely related to the safety issues that had been plaguing its experimental products for years.
The company’s preliminary data, which was promoted in yet another bid to keep investors from leaving, also included the caveat that Moderna had decided to pause trials for this particular product, which was a single-injection mRNA treatment for the chikungunya virus. That treatment was being developed in partnership with the Pentagon’s DARPA. Other more positive data from a preliminary trial were also released at this meeting. That trial, however, was for an mRNA treatment for cytomegalovirus, “a common virus that is usually kept in check by the body’s immune system and rarely causes problems in healthy people,” meaning its mRNA vaccine for that condition was unlikely to ever be lucrative.
Not long after this lackluster investors meeting, on September 26, 2019, the once highly secretive Moderna announced it would collaborate with researchers at Harvard University “in hopes that the research will spur new drugs,” as its product pipeline appeared to have stalled. Moderna president Stephen Hoge described the collaboration as select Harvard researchers receiving “a package of stuff that we put our blood, sweat, and tears in, and then someone’s going to do something with it. We’ll find out afterward how that went.” For a company long known for its extreme secrecy in an already secretive industry, Moderna’s arrangement with Harvard, which it admitted was “unusual,” came across as somewhat desperate.
A month later, at the 2019 Milken Institute Future of Health Summit, there was a panel discussion on universal flu vaccines and how a “disruptive” event would be needed to upset the long-existing bureaucratic vaccine-approval process to facilitate wider adoption of “nontraditional” vaccines, such as those produced by Moderna. Panel speakers including former FDA commissioner Margaret Hamburg, a veteran of the 2001 Dark Winter exercise and scientific advisor to the Gates foundation, as well as Anthony Fauci of the National Institutes of Health’s National Institute of Allergy and Infectious Diseases (NIAID) and Rick Bright of BARDA, who previously worked for the Gates-funded PATH. The panel discussion notably took place shortly after the controversial coronavirus pandemic simulation called Event 201, whose moderators and sponsors had been intimately involved in 2001’s Dark Winter.

Screengrab from the 2019 Milken Institute Universal Flu Vaccine panel. Full video available here.
During the panel, the moderator—Michael Specter of the New Yorker—asked the question: “Why don’t we blow the system up? Obviously, we just can’t turn off the spigot on the system we have and then say ‘Hey! everyone in the world should get this new vaccine we haven’t given to anyone yet,’ but there must be some way.” Specter then mentioned how vaccine production is antiquated and asked how sufficient “disruption” could occur to prompt the modernization of the existing vaccination development and approval process. Hamburg responded first, saying that as a society we are behind where we need to be when it comes to moving toward a new, more technological approach and that it is now “time to act” to make that a reality.
Several minutes later, Anthony Fauci stated that the superior method of vaccine production involves “not growing the virus at all, but getting sequences, getting the appropriate protein and it sticking in on self-assembling nanoparticles,” essentially referring to mRNA vaccines. Fauci then stated: “The critical challenge . . . is that in order to make the transition from getting out of the tried and true egg-growing [method] . . . to something that has to be much better, you have to prove that this works and then you have got to go through all of the critical trials—phase 1, phase 2, phase 3—and show that this particular product is going to be good over a period of years. That alone, if it works perfectly, is going to take a decade.” Fauci later stated that there is a need to alter the public’s perception that the flu is not a serious disease in order to increase urgency and that it would be “difficult” to alter that perception along with the existing vaccine development and approval process unless the existing system takes the posture that “I don’t care what your perception is, we’re going to address the problem in a disruptive way and an iterative way.”
During the panel, Bright stated that “we need to move as quickly as possible and urgently as possible to get these technologies that address speed and effectiveness of the vaccine” before discussing how the White House Council of Economic Advisers had just issued a report emphasizing that prioritizing “fast” vaccines was paramount. Bright then added that a “mediocre and fast” vaccine was better than a “mediocre and slow” vaccine. He then said that we can make “better vaccines and make them faster” and that urgency and disruption were necessary to produce the targeted and accelerated development of one such vaccine. Later in the panel, Bright said the best way to “disrupt” the vaccine field in favor of “faster” vaccines would be the emergence of “an entity of excitement out there that’s completely disruptive, that’s not beholden to bureaucratic strings and processes.” He later very directly said that by “faster” vaccines he meant mRNA vaccines.
The Bright-led BARDA and the Fauci-led NIAID in just a few months’ time became the biggest backers of the Moderna COVID-19 vaccine, investing billions and co-developing the vaccine with the company, respectively. As will be explained in Part II of this series, the partnership between Moderna and the NIH to co-develop what would soon become Moderna’s COVID-19 vaccine was being forged as early as January 7, 2020, long before the official declaration of the COVID-19 crisis as a pandemic and before a vaccine was proclaimed as necessary by officials and other individuals. Not only did the COVID-19 vaccine quickly become the answer to nearly all Moderna’s woes but it also provided the disruptive scenario necessary to alter the public’s perceptions of what a vaccine is and eliminate existing safeguards and bureaucracy in vaccine approval. (Watch the 2019 Universal Flu Vaccine event here.)
As Part II of this series will show, it was an alleged mix of “serendipity and foresight” from Moderna’s Stéphane Bancel and the NIH’s Barney Graham that propelled Moderna to the front of the “Warp Speed” race for a COVID-19 vaccine. That partnership, along with the disruptive effect of the COVID-19 crisis, created the very “Hail Mary” for which Moderna had been desperately waiting since at least 2017 while also turning most of Moderna’s executive team into billionaires and multi-millionaires in a matter of months.
However, Moderna’s “Hail Mary” won’t last – that is, unless the mass administration of its COVID-19 vaccine becomes an annual affair for millions of people worldwide. Even though real-world data since its administration began challenges the need for as well as the safety and efficacy of its vaccine, Moderna – and its stakeholders – cannot afford to let this opportunity slip through fingers. To do so would mean the end of Moderna’s carefully constructed house of cards.
Author’s Note: Dr. Michael Palmer, Dr. Meryl Nass and Catherine Austin Fitts contributed much-appreciated feedback and guidance on this article. Special thanks to Katy M. for copy-editing help.
October 8, 2021
Posted by aletho |
Corruption, Deception, Science and Pseudo-Science, Timeless or most popular | COVID-19 Vaccine |
Leave a comment
The government has refused to release the minutes of the meeting in which its vaccine advisory committee decided not to recommend vaccinating all 12-15 year olds against covid-19.1
The UK Health Security Agency, which replaced Public Health England, rejected a freedom of information request for the document on the grounds that it intended to publish the minutes “in due course.”
The agency argued that it was in the public interest to withhold the information until it could be released in a “simultaneous, coordinated manner” and that disclosing the minutes before they were finalised could “result in a false impression of the contents of the meeting.” The decision is being appealed.
On 3 September the Joint Committee on Vaccination and Immunisation (JCVI) said that it would not be recommending universal vaccination for 12-15 year olds because although the health benefits of vaccination were “marginally greater than the potential known harms,” the margin of benefit was considered too small.2 The committee did not explain what factors its conclusion was based on, and neither the minutes nor the data behind the decision have been made public.3
The JCVI asked ministers to seek further advice from the UK’s chief medical officers on the wider potential benefits of vaccination. The government later (13 September) accepted the chief medical officers’ recommendation to vaccinate all 12-15 year olds on the basis of an assessment that included transmission in schools and the effect on children’s education.4
In a letter dated 5 October academics from Independent SAGE wrote to the JCVI highlighting the fact that, despite the committee’s own policy stating that draft minutes would be published within six weeks of each meeting, the last publicly available minutes were from February 2021.5
They urged the JCVI to “abide by its code of practice and be open and transparent through rapid publishing of all agendas, supporting papers and minutes,” arguing that “public confidence in vaccination programmes is assisted by clear and consistent processes and messaging.”
They added, “In that spirit, we wish to have a public assurance from JCVI that all future considerations of covid-19 vaccines, including the extension of vaccination to children under 12 years of age, will be conducted openly and transparently.”
Footnotes
- bmj.com View and Reviews—Helen Salisbury: Official hesitancy is not helping (BMJ 2021;374:n2366; doi:10.1136/bmj.n2366)
October 8, 2021
Posted by aletho |
Deception, Science and Pseudo-Science | COVID-19 Vaccine, UK |
Leave a comment
Americans may be under the illusion that the Hippocratic Oath compels doctors to do no harm to their patients. Unfortunately, that is not an ironclad injunction that protects patients from harm. There are several reasons for this. First, there is not a universal “Hippocratic Oath” to which all doctors must adhere. Indeed, according to the National Institutes of Health, the Hippocratic Oath is not required by most modern medical schools, and doesn’t expressly state “First, do no harm.” Actually, in the oath’s third paragraph, it does state the physician would do no harm. Leaving out the word “first” changes nothing. Interestingly, the oath’s fourth paragraph explicitly states, “I will not give a lethal drug to anyone if I am asked, nor will I advise such a plan; and similarly I will not give a woman a pessary to cause an abortion.”
Putting aside the idea of an oath sworn to mythical characters known as Greek gods and goddesses, and the subject of abortion, why would physicians even need to take any oath to not harm their patients? Up to current times, patients have held their doctors in the highest regard. It would not be a stretch to say people held their doctors and medical staff with inviolate trust at the center of their relationship. Sadly, that almost sacred relationship between doctor and patient has, over the past almost two years, been eroded and blown up by COVID-19 and the Left’s weaponizing of it. Every day we are learning more and more how this virus came about. However, the genesis of COVID-19 is not now the main concern of most people.
What concerns more and more people is the alarming and growing demand being pushed by corporate federal proxies for experimental gene therapies (aka “vaccines”) to be mandated for all Americans. To use the medical profession as a tyrannical tool against the most vulnerable is beyond unconscionable. There are some doctors and medical scientists pushing back and doing what the whole medical profession should be doing.
For full disclosure and trust in the vaccines, the main question is what are all the substances in the vaccines? Are data safety sheets for the Pfizer, Astra-Zeneca, Moderna, and Johnson & Johnson vaccines being provided to medical personnel and patients? If so, are 100% of the ingredients listed? What about the reported graphene oxide or aborted fetal cells in any of the vaccines? What about all the deaths and injuries caused by these vaccines? These are some of the many questions that must be fully addressed. There is something deadly going on and it is more than a virus with a cure rate of 99% for most people.
While the ugly politics play out, the medical profession must get back to basics and stand united against any and all efforts made to dissuade them from putting patients first. If a patient wants one of the vaccine shots, first, do no harm! Educate yourselves on what is in the vaccines. Then give patients all the information they need for informed consent for the shot. Medical personnel must stand against any order which may mandate all people have to get a COVID shot. Every person has the right to exercise his or her own free will and should never be coerced by government or anyone else to do or not do something that impacts their lives. Allowing medical tyranny to rule the day will have vast consequences for everyone. In Colorado, a woman in stage 5 renal failure is denied a transplant because neither she nor her donor was vaccinated against the COVID-19 virus. What a tragedy for this woman whose very life depends on a kidney transplant!
If medical professionals bow to any edict other than their own conscience and good medical practice, they should be shunned. We may need to start taking personal responsibility and making better decisions that affect our well-being. After all, we are the first responders to our own health!
October 8, 2021
Posted by aletho |
Deception, Science and Pseudo-Science, Timeless or most popular, War Crimes | COVID-19 Vaccine |
Leave a comment
A study published Sept. 30, in the peer-reviewed European Journal of Epidemiology Vaccines found “no discernible relationship” between the percentage of population fully vaccinated and new COVID cases.
In fact, the study found the most fully vaccinated nations had the highest number of new COVID cases, based on the researchers’ analysis of emerging data during a seven-day period in September.
The authors said the sole reliance on vaccination as a primary strategy to mitigate COVID-19 and its adverse consequences “needs to be re-examined,” especially considering the Delta (B.1.617.2) variant and the likelihood of future variants.
They wrote:
“Other pharmacological and non-pharmacological interventions may need to be put in place alongside increasing vaccination rates. Such course correction, especially with regards to the policy narrative, becomes paramount with emerging scientific evidence on real-world effectiveness of the vaccines.”
As part of the study, researchers investigated the relationship between the percentage of population fully vaccinated and new COVID cases across 68 countries and 2,947 U.S. counties that had second dose vaccine, and available COVID case data.
For seven days preceding Sept. 3, researchers computed COVID cases per one million people for each country, as well as the percentage of population that was fully vaccinated.

Relationship between cases per 1 million people (last 7 days) and percentage of population fully vaccinated across 68 countries as of September 3, 2021
Notably, Israel with more than 60% of its population fully vaccinated, had the highest COVID cases per 1 million people during the seven-day period.
Iceland and Portugal, with more than 75% of their populations fully vaccinated, had more COVID cases per 1 million people than countries such as Vietnam and South Africa, where only about 10% of the population is fully vaccinated.
Across U.S. counties, the median new COVID cases per 100,000 people during the seven-day period was similar across the categories of percentage of population fully vaccinated.

Percentage of counties that had an increase of cases between two consecutive 7-day time periods by percentage of population fully vaccinated across 2947 counties as of Sept. 2, 2021
The researchers found a substantial county variation in new COVID cases within categories of percentage of population fully vaccinated. There also appeared to be no significant signaling of COVID cases decreasing in counties where a higher percentages of the population was fully vaccinated.
Of the top five counties with the highest percentage of population fully vaccinated (99.9% – 84.3%), the Centers for Disease Control and Prevention (CDC) identified four as “high” transmission counties.
Three of the four counties classified as “high” transmission had fully vaccinated rates of 90% or higher. Conversely, of the 57 counties classified as “low” transmission by the CDC, 15 had fully vaccinated rates of 20% or lower.
The findings also showed no discernible association between COVID cases and fully vaccinated rates when a one-month lag was considered, to account for the 14-day period it takes for a vaccine to be considered effective.
The authors suggested a correction to the policy narrative is warranted, as increasing vaccination rates is not enough. “Such course correction, especially with regards to the policy narrative, becomes paramount with emerging scientific evidence on real-world effectiveness of the vaccines,” they wrote.
The authors cited data from the Ministry of Health in Israel showing the effectiveness of two doses of Pfizer’s COVID vaccine against preventing SARS-CoV-2 infection was reported to be 39% — substantially lower than the reported trial efficacy of 96%.
Emerging research also shows immunity derived from Pfizer’s COVID vaccine may not be as strong as natural immunity acquired through infection.
A substantial decline in immunity from mRNA vaccines six months post immunization has also been reported along with an increasing number of breakthrough cases among the fully vaccinated, the researchers said.
The authors said stigmatizing populations over vaccines can do more harm than good, and non-pharmacological prevention efforts need to be renewed in order to learn to live with COVID “in the same manner we continue to live 100 years later with various seasonal alterations of the 1918 Influenza virus.”
Breakthrough cases significantly underreported as FDA reviews booster data
The number of vaccinated people testing positive for COVID is on the rise, and doctors in Ohio are reporting more breakthrough cases across hospital systems.
However, only certain types of COVID breakthrough cases are reported at both the state and federal level, leaving patients with mild cases underreported.
“We estimate anywhere from two to 10 times as many positives that are being reported is the real situation,” said Dr. David Margolius, division director of internal medicine at MetroHealth in Cleveland.
“It’s still rare, but I get a dozen COVID positive cases a day in my basket, and usually three or four of them have been vaccinated,” said Margolius.
The Ohio Department of Health and the CDC only report breakthrough cases in patients requiring hospital admission, or cases that resulted in death.
The CDC said it made the change in May in order to “maximize the quality of data collected on cases of greatest clinical and public importance.”
As of Sept. 27, the CDC had received reports from 50 U.S. states and territories of 22,115 patients with COVID vaccine breakthrough infection who were hospitalized or died.
The CDC said the number of COVID vaccine breakthrough infections reported to the agency are an undercount of all SARS-CoV-2 infections among fully vaccinated persons, especially of asymptomatic or mild infections.
In addition, national surveillance relies on passive and voluntary reporting, and data are not complete or representative, according to the CDC.
Massachusetts health officials on Tuesday reported nearly 4,000 new breakthrough cases over the past week, and 46 more deaths, according to NBC Boston.
In the last week, 3,741 new breakthrough cases were reported, with 125 more vaccinated people hospitalized.
This brings the total number of breakthrough cases in Massachusetts to 40,464 — out of 4.63 million vaccinated people — and the death toll among people with breakthrough infections to 300.
According to the Vermont Daily Chronicle, which cited statistics from Vermont’s Department of Health, 76% of the state’s COVID fatalities in September were breakthrough cases, with just eight of the 33 Vermonters who died being unvaccinated.
As of Tuesday, 88% of all eligible Vermonters age 12 and over had been vaccinated with at least one shot.
Health Department spokesperson Ben Truman said most of the vaccine “breakthrough” fatalities were elderly. Because they were among the first vaccinated, Vermont’s elderly “have had more time to potentially become a vaccine breakthrough case,” he said.
According to The Washington Post, Dr. Peter Marks, director of the U.S. Food and Drug Administration’s (FDA) Center for Biologics Evaluation and Research, said Tuesday updated data might make a strong case in support of everyone 18 and older being eligible for COVID vaccine boosters, but the agency will have to see whether its outside advisers agree.
The remarks from Marks came during a webinar as the FDA prepares to meet Oct. 14 and 15 with its outside advisers to discuss authorizing Moderna and Johnson & Johnson COVID booster shots.
Megan Redshaw is a freelance reporter for The Defender. She has a background in political science, a law degree and extensive training in natural health.
© 2021 Children’s Health Defense, Inc. This work is reproduced and distributed with the permission of Children’s Health Defense, Inc. Want to learn more from Children’s Health Defense? Sign up for free news and updates from Robert F. Kennedy, Jr. and the Children’s Health Defense. Your donation will help to support us in our efforts.
October 8, 2021
Posted by aletho |
Science and Pseudo-Science | Covid-19, COVID-19 Vaccine, European Union, United States |
Leave a comment
GAINESVILLE, Florida – Florida Gov. Ron DeSantis announced on Friday that the city of Gainesville has withdrawn a COVID-19 vaccine mandate for workers after pressure from his administration and a court ruling against the policy last month.
“We are not going to allow our first responders and government employees, many of whom have been on the front lines for over a year and a half, to be cast aside by local politicians’ mandates,” DeSantis said in a press release. “This reversal by the City of Gainesville is a victory for liberty.”
According to the press release, the Florida Department of Health (FDOH) received a letter from Gainesville last week informing the department that city commissioners “voided” a recent vaccination mandate for all public employees.
The FDOH had written to Gainesville mayor Lauren Poe days earlier, threatening to enforce a law signed by DeSantis in May that bans government entities, schools, and businesses from demanding COVID vaccination status. The law, S.B. 2006, allows up to $5,000 in fines per violation.
Gainesville told the health department in response that there are now “zero employees who are subject to a City COVID-19 employer vaccination requirement.”
“Please be advised that on September 23, 2021, the City Commission of the City of Gainesville took action to rescind their August 5th action related to COVID-19,” city manager Lee Feldman wrote, adding that the vaccine rules “have been voided.”
The day before the FDOH notice, a Florida judge temporarily halted Gainesville’s vaccine mandate following a lawsuit brought by city workers and backed by the DeSantis administration.
“The city failed to put on any evidence that the Vaccine Mandate serves a compelling state interest or that the Vaccine Mandate was the least restrictive means to accomplish that interest,” Circuit Judge Monica Brasington ruled September 22. Gainesville, in fact, “did not put on any evidence, at all, at the injunction hearing,” she noted.
The City Commission of Gainesville voted to rescind the rule the next day.
More than 200 city workers, including police officers, firefighters, and other first responders, sued to block the mandate, with the backing of Florida Attorney General Ashley Moody, a Republican who filed a brief in support of the workers in September.
Last month, Gov. DeSantis announced that Florida would move to penalize violations of the state’s ban on COVID-19 vaccine requirements, such as those in Gainesville, with “millions of dollars” in fines.
“If a government agency in the state of Florida forces a vaccine as a condition to employment, that violates Florida law, and you will face a $5,000 fine for every single violation,” the governor declared at a press conference near Gainesville. “If you look at places here in Alachua County, like the city of Gainesville, I mean, that’s millions and millions of dollars potentially in fines.”
“We will certainly move to impose the fines as the law requires when we see people that are denied access,” he said.
The DeSantis administration continues to fight local vaccine mandates in multiple counties, including Leon County, where city officials confirmed this weekend that they fired 14 employees who refused to disclose their vaccination status by Friday, in violation of S.B. 2006.
The FDOH had warned the county last week that it must “immediately rescind” the coercive vaccine policy and “refrain from terminating any employees who decline to produce proof of vaccination.”
“Fines may be assessed based on each employee who was required to submit proof of vaccination as a condition of continued employment,” wrote Doug Woodlief, FDOH division director for Emergency Preparedness and Community Support. Those fines could cost Leon County over $3.5 million, according to the Tallahassee Democrat.
Orange County also faces potential fines due to an ongoing COVID vaccine mandate, which prompted a lawsuit from dozens of firefighters last week, local news reported. The county downgraded the mandate days after Gov. DeSantis’ press conference last month, but has harassed unvaccinated employees and impeded them from being promoted, firefighters have said.
The Gainesville workers’ victory against compulsory COVID vaccination follows similar recent successes in other states. In Arizona, the city of Tucson backed down on a vaccine mandate last month after Republican Attorney General Mark Brnovich threatened to withhold up to $175 million in funding from the city, citing state laws enacted by Republicans earlier this year.
October 7, 2021
Posted by aletho |
Civil Liberties, Science and Pseudo-Science | Covid-19, COVID-19 Vaccine, United States |
Leave a comment
Three US medical certifying boards have warned doctors that they risk losing their certification and licence if they spread covid vaccine misinformation.
Internists, family doctors, and paediatricians received an email on 9 September that quoted a warning from the Federation of State Medical Boards in July1 which read: “Providing misinformation about the covid-19 vaccine contradicts physicians’ ethical and professional responsibilities, and therefore may subject a physician to disciplinary actions, including suspension or revocation of their medical licence.”2
Richard Baron, president and chief executive of the American Board of Internal Medicine (ABIM), told The BMJ that the move was an attempt to establish a standard of care. “As standard setting organisations, we thought it was important to be on record, in a public way, to make clear that putting out flagrant misinformation is unethical and dangerous during a pandemic.” Baron said that the statement has been well received—“4 to 1 positive.” But community physicians contacted by The BMJ thought differently.
“When I got that email I thought I’d better not put anything on social media about vaccines,” said Shveta Raju, a community physician in the Atlanta, Georgia, area, who has treated covid patients and led the vaccination effort at her outpatient clinic.
“The email was sent more as a veiled threat to keep doctors on the official, established narrative, and that’s what I find chilling,” said a paediatrician who pseudonymously blogs under the name Elizabeth Bennett. “Pandemic or no, there is a problem with having an ill defined concept of misinformation that’s tied to public health messaging that hasn’t been consistent. How are physicians supposed to figure out what is misinformation when public health messaging swings so wildly?” Bennett asked.
Undefined offence
Baron said that the statement was also intended to signal the certifying boards’ support for physicians “trying to do the right thing.”
“We wanted to support that group and say ‘hey, we do have a standard of care here and you are doing the right thing when you uphold it,’” he said.
Raju responded, “If that was their intent, they should have defined misinformation. By leaving it undefined, the message was that we can’t talk about this at all.” She said that physicians are, by and large, a conservative group. “If they’re not sure what can be deemed misinformation, physicians would rather be quiet.”
Bennett concurred: “The thing I find most alarming is that they don’t define misinformation, but if they strip you of your board certification, you would lose your means of earning a living.”
Doctors spreading misinformation?
Official and social media company efforts to target “vaccine misinformation” predate the pandemic.3 But the new statement from ABIM, the American Board of Family Medicine, and the American Board of Paediatrics is one of several recent statements putting doctors in the spotlight for the first time.
In Canada, warnings about physician information began earlier, when in April the College of Physicians and Surgeons of Ontario declared that physicians “have a professional responsibility not to communicate anti-vaccine, anti-masking, anti-distancing, and anti-lockdown statements or promote unsupported, unproven treatments for covid-19.”4
The Canadian statement triggered an outcry, leading to a clarification that the statement was “not intended to stifle a healthy public debate about how best to address aspects of the pandemic.” But concerns continued. In June, a Canadian member of parliament held a press conference on censorship of Canadian clinicians and scientists. YouTube removed the video of the meeting.56
The BMJ asked ABIM about the size of the problem of board certified physicians spreading misinformation.
“We don’t have a sense of numbers of physicians spreading misinformation,” Baron said. “We’re at the beginning.” He believed it was only a “small number of doctors.” The medical boards opted to send the statement to all doctors, he said, because focusing on just the offending individuals would “miss the impact they’re having because of how much their voices are being amplified.”
As an example of “unprofessional or unethical behaviour,” Baron cited the case of a Florida doctor offering medical exemptions from mask wearing for $50 (£37; €43).7
Personalised medicine—or one-size-fits-all?
The BMJ asked whether physicians expressing doubt about the need for booster doses or vaccination of patients with natural immunity—two matters that have been the subject of debate and changing official guidance—would qualify as misinformation.8 “I don’t think we have concerns with doctors wrestling with areas where the science is unclear,” Baron said, “but there is no debate about whether people should get a primary vaccination series.”
Raju worries about the impact on personalised care. “The job of physicians is to take guidelines and apply them to the patient in front of them.” But now “physicians are basically being told that when it comes to covid vaccines it’s one-size-fits-all.”
Baron said, “We’re not trying to stifle conversations between doctors and patients. We understand that different people may look at evidence in different ways, but when you have an overwhelming preponderance of medical consensus in a certain area, you need at least to tell patients that there is an overwhelming professional consensus here.”
Cautious approach
Jeffrey Flier, former dean of Harvard Medical School, said that in the context of the pandemic, he was “not opposed to certain levels of misinformation triggering a decision to question somebody’s licence.” He said, “I can see this being an appropriate remedy at a time of public health emergency.
“But this is not how the system for licensure and certification has traditionally worked, and creates many opportunities for mistaken judgment about what is and is not misinformation, and those decisions would have to be rendered with extreme caution.”
Flier added, “We have to remember that there are legitimate areas of debate, and such matters should not fall within the scope of disciplinary actions.”
“There are reasons to be concerned that state boards might be unprepared for these kinds of decisions at a time when so many aspects of covid policy have been enmeshed with political views.”
Footnotes
-
This article was updated on 4 October to make clear that it was medical certifying boards, rather than licensing boards, that emailed physicians. The email quoted an earlier warning from the Federation of State Medical Boards.
-
Competing interests: PD gave a public statement at a 17 September 2021 FDA advisory committee to discuss covid-19 vaccines, where he highlighted the joint statement. The views and opinions expressed here are those of the author and do not necessarily reflect official policy or position of the University of Maryland.
-
Provenance: commissioned; not externally peer reviewed.
This article is made freely available for use in accordance with BMJ’s website terms and conditions for the duration of the covid-19 pandemic or until otherwise determined by BMJ. You may use, download and print the article for any lawful, non-commercial purpose (including text and data mining) provided that all copyright notices and trade marks are retained.
https://bmj.com/coronavirus/usage
View Abstract
October 7, 2021
Posted by aletho |
Deception, Full Spectrum Dominance, Science and Pseudo-Science, War Crimes | Canada, Covid-19, COVID-19 Vaccine, Human rights, United States |
Leave a comment

Dr. Rochagné Kilian
OWEN SOUND, Ontario – Dr. Rochagné Kilian recently resigned as an emergency room and family practice physician due to her concerns that the Ontario health system and Grey Bruce Health Services (GBHS) crossed ethical lines throughout the pandemic.
In a virtual meeting that included GBHS CEO Gary Sims and other staff members, Dr. Kilian asked Sims a series of questions about what she believes is unethical behaviour on behalf of the Ontario health system at all levels. Sims appeared to be unprepared for difficult questions pertaining to the ongoing rollout of vaccination mandates and vaccine segregation restrictions the Ontario heath system is championing.
Kilian estimated that 80 percent of the patients she saw in the ER during the past month who had inexplicable symptoms were “double vaxxed.”
Dr. Kilian relocated to Owen Sound – a small city in Grey County, Ontario – from South Africa after previously working in British Columbia. When she resettled in Owen Sound with her family, she expressed to a local paper how happy she was to live there: “Our recruitment to Owen Sound might have been by chance, but our choice to settle here was definitely not. Our four months in Owen Sound have been blessed. A little town with lots of soul, surrounded by beautiful landscapes, filled with welcoming residents and businesses, and exciting festivals, programs and activities. We truly feel fortunate to raise a family here.”
The first issue that Dr. Kilian brought up during the meeting was informed consent regarding the COVID jab and what she considered to be a coercive mentality of pressuring people to accept medications that she pointed out are still in “clinical trials.”
An GBHS administrator did not answer her question directly, but instead passed the buck to the provincial government and stated they do not have “oversight or input” regarding consent mechanisms presented to patients.
Kilian added that having more input into what patients are consenting to is something that GBHS “should consider,” especially in light of enacting the government-recommended vaccination mandates with their own staff.
Referring to informed consent and mandating experimental vaccines that been linked to thousands of deaths and injuries, Sims explained that because of the “pandemic,” certain procedural normalities will not take place.
“In a pandemic, some of those pieces that you think would be there [mechanisms of informed consent from the government] when you have lots of time to review stuff … in a pandemic, they’re going to pass mandates, and they’re going to pass laws, and they’re going to pass directives as needed to manage that pandemic,” he said. “And some of the things … will feel like they’re infringing on or taking short cuts … they are doing that directly to save lives.”
Sims then stated that there is a “massive scientific group” that has reviewed all the issues pertaining to the vaccines and their usage and rollout. This meeting took place August 23. Since then, the Ontario government has admitted that at least one of the vaccines pushed on the public has caused serious heart problems in young men, who are at virtually no statistical risk of suffering severely from COVID.
Dr. Kilian pressed Sims about claims that protocols of informed consent can be skirted due to an emergency, and clarified that the Tri-Council Policy Statement stipulates that an emergency situation does not warrant skirting protocols that protect the population from being put at risk due to medical experimentation. The Tri-Council Policy Statement is a Canadian guideline for the ethical conduct of research involving humans and/or human biological materials. As the vaccinations are still technically under experimental trial, they are being implemented under a research-based framework on the population.
It was Kilian’s opinion that the ethical framework is being ignored, thus health workers and citizens are being forced to take something against their will that is not proven to be safe or effective in the long term, as a result of vaccination mandates.
Sims reacted sharply to Kilian and said, “Nobody is forcing you to do this, you have a right to say no, but the reality is the government has the right to say that you’re not employed.”
“When the law looks at it, the law is saying you have the right to do it [enforcing vaccine mandates],” he added.
Dr. Byram Bridle, a University of Guelph professor, recently released a letter he sent to the president of his university that called into question the legitimacy of vaccine mandates, both from a medical and legal perspective. He stated in the letter that he is “confident there will be lawyers willing to test this in court.”
Dr. Kilian asked a final question in the virtual meeting about the claims that Sims and others at GBHS have made about the majority of COVID-associated cases in the region being among the unvaccinated. She asked if there was a detailed database that could be shared to prove this point. Sims stated that the vaccination status of the individuals who have been admitted in his region could not be released due to privacy reasons, but that the provincial government would have the information.
He then claimed that provincially, “less than 0.7 percent of people who ended up in ICUs were vaccinated.”
He referred to the “third wave” of COVID in Ontario that he said was due to the Delta variant, and stated that “it was all unvaccinated” who fell seriously ill at that time. The third wave in Ontario is reported to have happened in April and May. The vast majority of Ontarians had not received their second dose of any COVID jab by that point, and the province has made it clear in numerous places that a person only counts as “full vaccinated” for clinical purposes after 14 days have passed since they have received their second dose.
During the month of May in the Grey Bruce region, there were a total three confirmed hospitalizations with COVID-19. Five people in the region died that month with a COVID-positive diagnosis, and two of the deaths were residents who died outside of the county.
Sims said on the call that a minimum “80 percent” and up to “97 percent” of ICU patients with COVID across the province were unvaccinated. It is impossible to reach that number given the Grey Bruce statistics because there are too few people for calculations to be mathematical doable.
He then intimated that there will be great fears among pediatric physicians regarding autumn COVID numbers “if children start to die.” There is no evidence to suggest that COVID is dangerous to children in any statistically significant way.
Dr. Kilian resigned from her position while on the call with GBHS administration and spoke about her situation on the The Strong and Free TruthCast, where she criticized the state of health care in Canada. She expressed that care for the individual patient has gone by the wayside during the “farce that we have been living through.”
She said that throughout the entire time that the pandemic has been declared, she has only admitted two patients to the ICU that tested positive for COVID. She then clarified that this did not mean they were in the ICU due to COVID, but only that they had tested positive. She stated that her emergency department was “dead” throughout all of the declared waves of COVID, and that she took pictures of the official numbers to prove that they “had nothing to do” with lack of patients.
Dr. Kilian added that since the rollout of COVID jabs, she has seen a striking uptick in patients who have been admitted with heart issues and do not fit risk categories. She stated that as more and more people have received the jab, she has seen a host of strange events in her patients. She spoke of “people coming with newly diagnosed high blood-pressure, diabetics that was controlled that are no longer controlled – their sugars are either through the roof or they’re down in the ground … The only factor … constant that changed in their life was the injection of an experimental biologic.”
October 7, 2021
Posted by aletho |
Civil Liberties, War Crimes | Canada, Covid-19, COVID-19 Vaccine, Human rights |
Leave a comment

A study appeared in the Lancet this week confirming that vaccine effectiveness against infection is fading fast.
The study involved 3,436,957 people over the age of 12 who are members of the healthcare organisation Kaiser Permanente Southern California. It sought to assess the effectiveness of the Pfizer vaccine against SARS-CoV-2 infections and COVID-19-related hospital admissions for up to six months, with a study period covering December 14th, 2020, to August 8th, 2021.
Comparing fully vaccinated to unvaccinated, and controlling for confounders such as prior infection, the researchers found that effectiveness against infection plummeted from 88% (95% confidence interval 86-89%) during the first month after double-vaccination to 47% (43-51%) after five months. The variation by age (depicted above) was largely within the margins of error.
Among sequenced infections, the researchers found vaccine effectiveness against Delta infection was 93% (85-97%) during the first month after double-vaccination but dropped to 53% (39-65%) after four months. Effectiveness against infection from other variants the first month after double-vaccination was 97% (95-99%), but declined to 67% (45-80%) at 4-5 months.

Pfizer vaccine effectiveness by Covid variant
Vaccine effectiveness against hospital admissions for Delta infection held up at around 93% (84-96%) for the six months across all ages.

Pfizer vaccine effectiveness against Covid hospitalisation
However, the researchers note that the latest data from Israel “suggests that some reduction in effectiveness against hospital admissions has been observed among older people (65 years and over) roughly six months after receiving the second dose of [Pfizer]”.
One question that’s arisen recently is to what extent vaccine effectiveness estimates are affected by whether more people who have been previously infected decide not to be vaccinated. According to this study the answer is: not very much at all. Among the unvaccinated, 2.3% had one or more previous positive PCR tests, only slightly more than the 2% of the double-vaccinated who did.
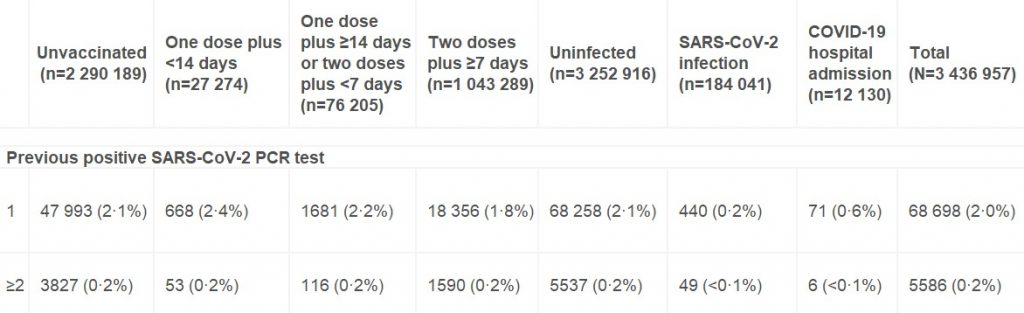
Number and percentage of individuals with one or more previous positive PCR tests
It’s also worth noting that although this study adjusts its raw estimates for no fewer than 22 potential confounding variables, the adjusted figures differ very little from the unadjusted figures in almost all cases. This suggests that unadjusted estimates from large population samples are often a fair approximation in the absence of sophisticated statistical analysis.
Given that the adjusted figures were little different to the unadjusted figures, however, it’s not immediately clear why the vaccine effectiveness estimates in this study, while low and declining, are so much higher than the latest unadjusted estimates derived from Public Health England data (namely, negative vaccine effectiveness in the over-40s, including minus-66% in those in their 40s). It doesn’t appear to be merely a matter of additional time elapsing, as most people in the U.K. weren’t double vaccinated until April, May or June, meaning only four or five months have elapsed until September, the same time period as in the study.
Could it be because the study period ended on August 8th, when the Delta surge in California was just getting going (see below)?
In the U.K. the vaccine effectiveness didn’t plunge until the second half of the Delta surge, the first part being dominated by infections in the unvaccinated (for reasons still not entirely clear). Did the new study finish too early to see the dramatic effect we’ve seen in England?
The authors say their study indicates that the decline in vaccine effectiveness is primarily a function of time rather than variant-related. However, the evidence from England would suggest otherwise, as in the same period of time, but later in the Delta surge, the decline has been far greater.
The decline in vaccine effectiveness in England was confirmed last week in a new Government-funded study (not yet peer-reviewed), which found that the reduction in transmission “declined over time since second vaccination, for Delta reaching similar levels to unvaccinated individuals by 12 weeks for [the AstraZeneca vaccine] and attenuating substantially for [Pfizer]”. In other words, within just three months AstraZeneca did nothing to prevent transmission, and Pfizer was scarcely better.
One of the main recommendations of the authors of both studies in light of their findings is for regular booster jabs – in the case of the first, where many of the authors are employees of and investors in Pfizer, this may be deemed hardly surprising. However, if effectiveness against serious disease is holding up, why give people boosters just to stop them getting and spreading what is effectively a cold, and which bestows more robust immunity as it goes? Furthermore, if the effectiveness declines after as little as three months, is it even possible to deliver enough boosters to have any impact on infection and transmission? Would it not be much better to say that the vaccines, by offering personal protection from serious disease to those who want it, have done their job? Better to move on and abandon any ideas of vaccine passports and mandates and boosters, and in general the now almost wholly pointless obsession with Covid vaccines.
October 7, 2021
Posted by aletho |
Science and Pseudo-Science | COVID-19 Vaccine |
Leave a comment
















